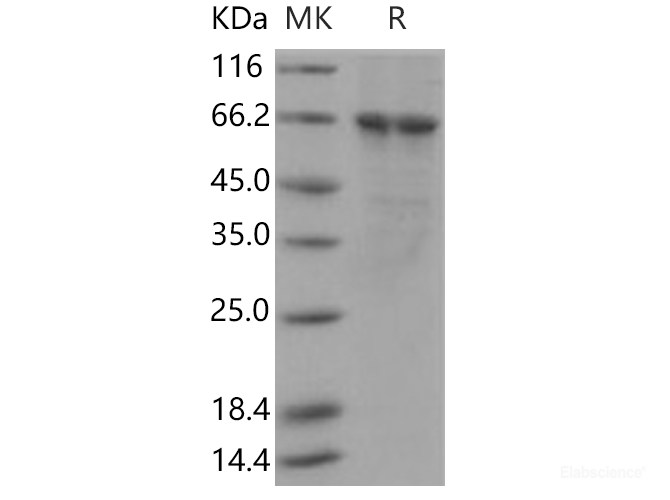
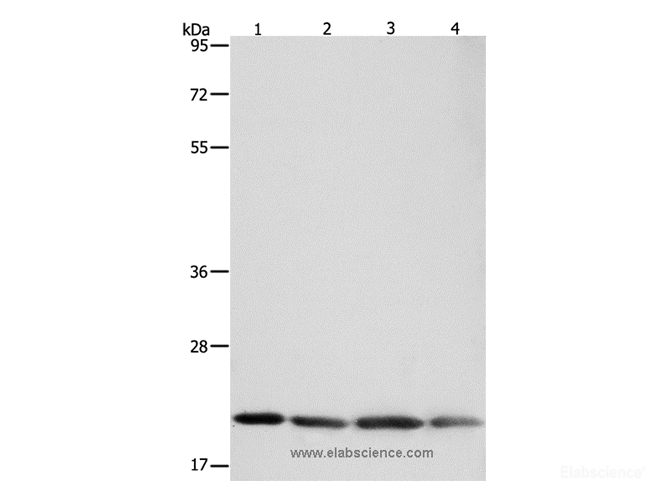
Background
Serine / threonine-protein kinase PLK1 / PLK-1, also known as polo-like kinase 1 (PLK-1) or serine / threonine-protein kinase 13 (STPK13), Polo-like kinases (PLKs), is a family of four serine / threonine protein kinases that are critical regulators of cell cycle progression, mitosis, cytokinesis, and the DNA damage response. PLK1 / PLK-1 is ubiquitously expressed. The mRNA and protein expression of PLK1 / PLK-1, -2 and -4 are coordinately regulated during cell cycle progression, but PLK3 levels are independent of the other three family members. PLK1 / PLK-1 is the most well characterized member of this family and strongly promotes the progression of cells through mitosis. During the various stages of mitosis PLK1 / PLK-1 localizes to the centrosomes, kinetochores and central spindle. Serine / threonine-protein kinase that performs several important functions throughout M phase of the cell cycle, including the regulation of centrosome maturation and spindle assembly, the removal of cohesins from chromosome arms, the inactivation of APC / C inhibitors, and the regulation of mitotic exit and cytokinesis. It is required for recovery after DNA damage checkpoint and entry into mitosis. PLK1 / PLK-1 is required for kinetochore localization of BUB1B, spindle pole localization of isoform 3 of SGOL1 and plays a role in regulating its centriole cohesion function. PLK1 / PLK-1 Phosphorylates BORA, and thereby promotes the degradation of BORA. PLK1 / PLK-1 also contributes to the regulation of AURKA function and phosphorylates SGOL1.