Caspase-mediated apoptosis is a fundamental biological process essential for development and tissue homeostasis. This process depends on a family of cysteine-aspartic proteases known as caspases. These enzymes are synthesized as inactive zymogens or pro-caspases. They achieve functional activation through proteolytic cleavage in response to specific apoptotic signals and initiate a hierarchical cascade to dismantle damaged cells. The detection of activated caspases supports in-depth apoptosis research and therapeutic efficacy evaluation. Common targets include executioner caspases such as caspase-3 and caspase-7, as well as initiator caspases such as caspase-8 and caspase-9. Such detection holds particular research value in oncology.
This review systematically summarizes the caspase family and its core functions in apoptosis. It further discusses the crosstalk between caspase-related cascades and inflammatory signaling pathways including pyroptosis and inflammasome activation. The article also illustrates the activation mechanisms of caspase‑3/7 in intrinsic and extrinsic apoptotic pathways. It introduces mainstream methods that apply fluorescent probes to detect caspase‑3 activation. Moreover, this paper emphasizes the biological function of caspase‑8 in extrinsic apoptosis and death receptor-mediated signaling. Finally, it clarifies the regulatory status of caspase‑9 within the mitochondrial intrinsic apoptosis pathway.
Table of Contents
1. Overview of caspase family and their role in apoptosis
2. Crosstalk between caspases and inflammatory signaling (e.g., pyroptosis, inflammasome)
3. Caspase-3/7 activation in intrinsic and extrinsic apoptosis pathways
4. Detection of caspase-3 activation using fluorescent probes
5. Role of caspase-8 in extrinsic apoptosis and death receptor signaling
6. Caspase-9 involvement in mitochondrial (intrinsic) apoptosis pathway
01 Overview of caspase family and their role in apoptosis
Caspases are a family of cysteine-aspartic proteases that exert indispensable functions in programmed apoptotic cell death, inflammatory responses, and diverse physiological cellular processes[1]. These proteases are evolutionarily highly conserved across species ranging from Caenorhabditis elegans to humans, thereby sustaining embryonic development and tissue homeostasis. Synthesized as inactive zymogens termed pro-caspases, caspases undergo site-specific proteolytic cleavage upon apoptotic stimuli to attain full enzymatic activity[1,2].
Accordingly, the caspase family is functionally divided into two major subgroups: initiator caspases and executioner caspases, which sequentially assemble a hierarchical signaling cascade to drive apoptotic progression.
Initiator caspases, comprising caspase-2, -8, -9 and -10, reside at the upstream apex of apoptotic cascades. Specifically, they recognize and converge pro-apoptotic signals derived from extracellular extrinsic pathways and intracellular intrinsic stress pathways. Moreover, these caspases harbor lengthy N-terminal prodomains containing canonical interaction motifs, including Caspase Recruitment Domain (CARD) and Death Effector Domain (DED)[3].
Notably, initiator caspase activation relies on an induced proximity model mediated by large scaffolding complexes. Once apoptotic signals induce multi-protein platform assembly, abundant pro-caspase monomers are recruited and concentrated locally. Consequently, forced molecular proximity facilitates caspase dimerization, which triggers conformational rearrangement and active site formation, followed by autocatalytic proteolytic maturation. Following activation, initiator caspases exhibit narrow substrate selectivity, and predominantly cleave and activate downstream executioner caspases[3].
By contrast, executioner caspases, including caspase-3, -6 and -7, act downstream in apoptotic signaling cascades. They feature short prodomains devoid of CARD or DED domains. Unlike initiator isoforms, executioner caspases cannot undergo spontaneous auto-activation; instead, they are directly processed and activated via proteolytic cleavage by upstream active initiator caspases, which removes inhibitory prodomains and exposes functional catalytic conformations[4].
Once activated, executioner caspases display broad substrate specificity toward hundreds of intracellular proteins. As a result, their coordinated proteolytic activity systematically dismantles cellular structures, generating canonical morphological and biochemical features of apoptosis, such as chromatin condensation, genomic DNA fragmentation, plasma membrane blebbing, and apoptotic body formation[3,4].
Although caspases are classically defined as core apoptotic executors, they additionally mediate versatile non-apoptotic events, including cell survival, proliferation, differentiation and inflammatory homeostasis. Furthermore, these biological functions rely on mild, transient and spatially restricted caspase activation rather than robust lytic activity. For example, caspases modulate neural development involving axonal guidance and synaptic maturation, as well as tissue remodeling during placental morphogenesis. In certain circumstances, caspases regulate signal transduction independent of cell death induction; for instance, caspase-dependent cleavage of sorting nexin 2 impairs retromer complex assembly and amplifies Met/hepatocyte growth factor receptor signaling. Meanwhile, inflammatory caspases (human caspase-1, -4, -5, -12; mouse caspase-1, -11, -12) operate independently from apoptotic caspase cascades, and primarily mediate maturation of pro-inflammatory cytokines such as Interleukin-1 beta (IL-1β) and Interleukin-18 (IL-18)[5].
Given their highly cytotoxic effects, caspase activation is subject to strict spatiotemporal regulation. Since caspases exist as inactive precursors under steady-state conditions, their maturation strictly depends on specific triggering signals. In addition, Inhibitors of Apoptosis Proteins (IAPs) serve as endogenous negative regulators that restrain caspase cascades and avoid aberrant premature activation[6].
Nevertheless, abnormal caspase homeostasis closely correlates with multiple pathological disorders. Inadequate apoptotic activity, caused by caspase inactivation or IAP overexpression, promotes tumorigenesis and autoimmune diseases. Conversely, excessive and ectopic caspase activation exacerbates neurodegenerative diseases, ischemic tissue injury and multiple inflammatory lesions. Accordingly, caspases have emerged as vital clinical therapeutic targets, and pharmacological modulation of caspase activity remains a hotspot in translational biomedical research.

Fig. 1 Mammalian caspase family and C. elegans caspase CED-3[2]. All mammalian caspases are of human origin except for murine caspase-11 and -12, for which no human counterparts have been identified yet. Phylogenetic relationships are based on sequence similarity among the protease domains. Alternative names are listed in parentheses after each caspase. Dotted box, DED domains; wavy boxes, CARD domain. Substrate preferences at the P1 to P4 positions are indicated. Based on the substrate specificity, caspases are divided into three groups (indicated in parentheses)[2].
02 Crosstalk between caspases and inflammatory signaling (e.g., pyroptosis, inflammasome)
Caspases, a family of cysteine proteases, are pivotal regulators of both programmed cell death and inflammatory signaling, with intricate crosstalk existing between apoptotic and inflammatory pathways, notably pyroptosis and inflammasome activation. This complex interplay determines cell fate and immune responses.
2.1 Caspase-1 and Inflammasome-Mediated Pyroptosis
The inflammasome, a pivotal multiprotein complex in innate immunity, is stimulated by pathogen-associated molecular patterns (PAMPs) and danger-associated molecular patterns (DAMPs). Subsequently, it initiates canonical caspase-1 and non-canonical human caspase-4/5 or mouse caspase-11 signaling cascades through a tightly controlled two-signal activation model: the first signal relies on Toll-like receptor (TLR)-mediated priming, whereas the second signal serves as direct activation cues. Once activated, caspase-1 processes pro-interleukin-1β (pro-IL-1β) and pro-interleukin-18 (pro-IL-18) into mature inflammatory cytokines to amplify inflammatory reactions. Meanwhile, caspase-1 also cleaves Gasdermin D (GSDMD) to trigger pyroptosis; the released GSDMD-N domains form membrane pores and induce lytic programmed cell death. Although pyroptosis exerts robust anti-intracellular pathogen immunity, aberrant activation thereof closely participates in the pathogenesis of diverse inflammatory diseases[7].
2.2 Crosstalk between Inflammatory and Apoptotic Caspases
Inflammatory caspases (caspase-1, -4, -5, -11) dominate pyroptosis whereas apoptotic caspases (caspase-3, -6, -7) govern classical apoptosis, yet extensive regulatory crosstalk interconnects these cascades. As a core extrinsic apoptotic initiator, caspase-8 bifunctionally modulates pyroptosis and inflammasome signaling: it switches cell death toward apoptosis upon Gasdermin D (GSDMD) deletion or caspase-1 suppression and bridges apoptotic and pyroptotic pathways via NOD-like receptor family pyrin domain containing 3 (NLRP3) regulation and GSDMD cleavage. Meanwhile, apoptotic executioners caspase-3 and -7 restrict pyroptotic progression as molecular brakes, as caspase-3 cleaves Gasdermin E (GSDME) to trigger pyroptosis-like cell death and caspase-7 inactivates Gasdermin B (GSDMB) to suppress non-canonical pyroptosis (which can be analyzed by the caspase 3/7 activity assay). Additionally, canonical apoptotic caspase-6 participates in Z-DNA binding protein 1 (ZBP1)-NLRP3 inflammasome activation, drives PANoptosis, and mediates host defense against Influenza A Virus (IAV), revealing widespread functional overlap between apoptotic and inflammatory caspase-mediated cell death and immune responses[8].
The interaction between different forms of programmed cell death, such as pyroptosis, apoptosis, and necroptosis, is complex and highly context-dependent. These pathways are not isolated but rather form an interconnected network, allowing for crosstalk and coordinated responses. The specific caspases involved ultimately determine the nature of the cell death and its immunological consequences. Understanding this intricate crosstalk is critical for developing targeted therapies for inflammatory diseases and cancers in which inflammasome activation and pyroptosis play significant roles.
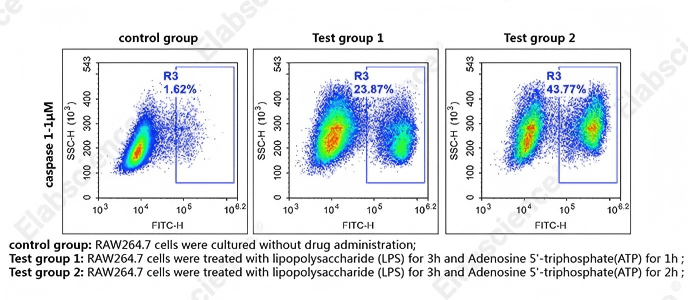
Fig. 2 Detection of caspase-1 activity in RAW264.7 cells. RAW264.7 cells were cultured with LPS for 3 h and ATP for 1 h (Test group 1), or with LPS for 3 h and ATP for 2 h (Test group 2), or without drug administration (control group). Cells were then stained with caspase-1 substrates (cat. no. E-CK-A481) and analyzed by flow cytometry. (The data are provided by Elabscience.)
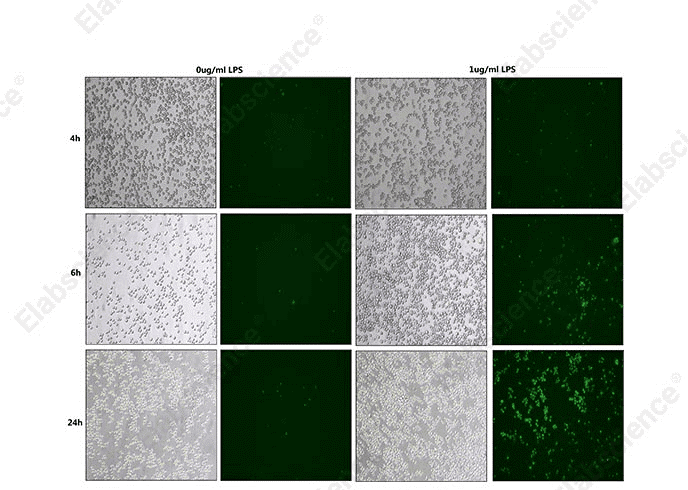
Fig. 3 Detection of caspase-1 activity in THP-1 cells. THP-1 cells were treated with LPS at concentrations of 0 μg/mL and 1 μg/mL. Caspase-1 substrates (cat. no. E-CK-A481) were then added, and the dynamic changes of caspase-1 activity were detected at 4 h, 6 h, and 24 h post-induction. (The data are provided by Elabscience.)
03 Caspase-3/7 activation in intrinsic and extrinsic apoptosis pathways
Caspase-3 and caspase-7 are pivotal executioner caspases downstream of initiator caspases in both intrinsic and extrinsic apoptotic cascades, which mediate cellular disassembly during programmed cell death. Although both proteases drive canonical morphological and biochemical alterations of apoptosis, they exhibit subtle distinctions in activation modes and pathway-specific functions.
In the intrinsic mitochondrial apoptotic pathway, diverse intracellular stresses including DNA damage, growth factor deprivation, and endoplasmic reticulum stress induce mitochondrial outer membrane permeabilization (MOMP). Subsequently, MOMP triggers the cytosolic release of pro-apoptotic molecules, predominantly cytochrome c. Upon binding to Apoptotic protease activating factor-1 (Apaf-1), cytochrome c assembles a wheel-shaped heptameric apoptosome complex in an ATP/dATP-dependent manner. Via CARD–CARD homotypic interactions, the apoptosome recruits procaspase-9 and promotes its dimerization and autocatalytic activation. Activated caspase-9 subsequently cleaves inactive zymogens procaspase-3 and procaspase-7, thereby converting them into mature functional enzymes that orchestrate downstream apoptotic execution[9].
By contrast, the extrinsic apoptotic pathway is triggered by extracellular death ligands, such as Fas Ligand (FasL) and Tumor Necrosis Factor alpha (TNFα), which bind to surface death receptors including Fas and Tumor Necrosis Factor Receptor 1 (TNFR1). This ligand-receptor engagement recruits adaptor protein Fas-Associated Death Domain (FADD) to assemble the Death-Inducing Signaling Complex (DISC). Within the DISC, concentrated procaspase-8 undergoes dimerization and spontaneous auto-activation. Active caspase-8 directly processes procaspase-3 and procaspase-7 to initiate terminal apoptotic cell demolition. Furthermore, in Type II cells, caspase-8 cleaves Bid protein to amplify apoptotic signaling; truncated Bid translocates to mitochondria and accelerates cytochrome c release, consequently crosstalking with the intrinsic pathway and forming a mitochondrial amplification loop that sustains caspase-9, caspase-3 and caspase-7 activation[10].
Once activated, caspase-3 and caspase-7 display broad substrate specificity toward hundreds of intracellular proteins, which can be analyzed by the caspase 3/7 activity assay. As a result, their coordinated proteolytic cleavage drives systematic cellular breakdown and produces hallmark apoptotic phenotypes, consisting of chromatin condensation, genomic DNA fragmentation, plasma membrane blebbing and apoptotic body formation. Among them, caspase-3 serves as the dominant apoptotic executor, targeting numerous structural and regulatory substrates such as nuclear lamins, cytoskeletal proteins (actin, gelsolin) and Inhibitor of Caspase-Activated DNase (ICAD). Cleavage of ICAD releases active Caspase-Activated DNase (CAD), which subsequently induces typical internucleosomal DNA ladder fragmentation (can be detected using the TUNEL assay kit). Accordingly, detection of caspase-3 activation serves as a vital biomarker for assessing early anti-tumor therapeutic efficacy[11].
Given the highly cytotoxic nature of caspases, their enzymatic activity undergoes stringent spatiotemporal control. Caspase-3 and caspase-7 are initially synthesized as inactive precursors, and their maturation strictly relies on initiator caspase-mediated proteolytic removal of inhibitory prodomains. Meanwhile, Inhibitors of Apoptosis Proteins (IAPs) function as endogenous negative regulators that suppress aberrant and premature caspase cascade activation. Aberrant caspase-3 homeostasis is closely associated with multiple disorders, encompassing malignancies, neurodegenerative diseases and immune deficiency disorders. Collectively, caspase-3 has become a core therapeutic target for natural and synthetic anti-cancer agents.
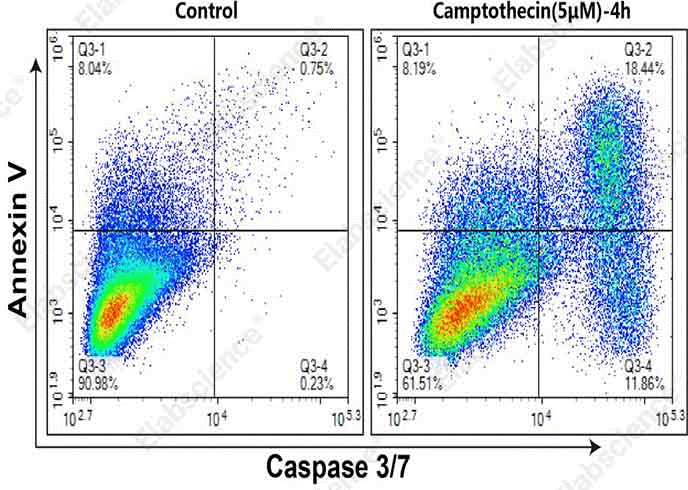
Fig. 4 Detection of caspase 3/7 activity and Annexin V in MOLT-4 cells. MOLT-4 cells were cultured without (left) or with (right) 5 μM Camptothecin for 4 h. Annexin V-APC single-positive cells (Q3-1) were early apoptotic cells with phosphatidylserine translocation. Annexin V and caspase-3/7 double-positive cells (Q3-2) were apoptotic cells with higher caspase-3/7 activity in the early to mid-late stage. Caspase-3/7 single-positive cells (Q3-4) were early apoptotic cells with caspase-3/7 activation. (The data are provided by Elabscience.)
Elabscience® Quick Overview of Popular Products:
Table 1. Reagents for Cell Apoptosis and Pyroptosis Research
|
Product Name |
Cat. No. |
|
Caspase 1 Activity Assay Kit(Colorimetric Method) |
E-CK-A381 |
|
Caspase 1 Activity Detection Substrate for Flow Cytometry |
E-CK-A481 |
|
Caspase 3/7 Activity Assay Kit(Colorimetric Method) |
E-CK-A383 |
|
Caspase 2 Activity Assay Kit(Colorimetric Method) |
E-CK-A382 |
|
Caspase 4 Activity Assay Kit(Colorimetric Method) |
E-CK-A384 |
|
Caspase 6 Activity Assay Kit(Colorimetric Method) |
E-CK-A386 |
|
Caspase 8 Activity Assay Kit(Colorimetric Method) |
E-CK-A388 |
|
Caspase 9 Activity Assay Kit(Colorimetric Method) |
E-CK-A389 |
|
Caspase 4 Activity Detection Substrate for Flow Cytometry |
E-CK-A484 |
|
Caspase 6 Activity Detection Substrate for Flow Cytometry |
E-CK-A486 |
|
Caspase 8 Activity Detection Substrate for Flow Cytometry |
E-CK-A488 |
|
Caspase 9 Activity Detection Substrate for Flow Cytometry |
E-CK-A489 |
|
Caspase 3/7 Activity Detection Substrate for Flow Cytometry |
E-CK-A483 |
|
Annexin V-FITC/PI Apoptosis Kit |
E-CK-A211 |
|
One-step TUNEL In Situ Apoptosis Kit (Green, FITC) |
E-CK-A320 |
|
One-step TUNEL Flow Cytometry Apoptosis Kit (Green, FITC) |
E-CK-A420 |
|
Caspase 3/7 and Annexin V Double Staining Apoptosis Kit |
E-CK-A831 |
04 Detection of caspase-3 activation using fluorescent probes
Detection of Caspase 3 is commonly performed using absorbance-based and fluorescence-based methods.Both fluorescence-based and absorbance-based (colorimetric) assays for caspase detection rely on the principle of enzymatic substrate hydrolysis, but they differ significantly in sensitivity, operational cost, and application scenarios, the detailed differences are shown in the table below.
Table 2. Comparison of fluorescence and absorbance methods for caspase-3 detection
|
Metric |
Fluorescence Method |
Absorbance (colorimetric) Method |
|
Sensitivity |
High, capable of detecting minute activity changes |
Lower, relies on color intensity, suitable for samples with higher enzyme activity |
|
Specificity |
High, precise substrate design with minimal background interference; detection of apoptosis in a single cell |
Moderate, may suffer from non-specific hydrolysis; detection of apoptosis in group cells |
|
Sample Requirement |
Requires only a small number of cells (0.5-2×105 cells/Test) |
Requires more cells or tissue lysate |
|
Instrumentation |
Requires a fluorescence microplate reader or fluorescence microscope or flow cytometer |
Can be performed with a standard spectrophotometer or microplate reader |
|
Application Scenarios |
High-throughput drug screening, dynamic monitoring, live-cell imaging |
Basic apoptosis detection in routine labs |
Elabscience® Caspase 3/7 Substrates (Green) are novel cell-membrane-permeable fluorogenic substrates applied for apoptosis detection across diverse cell models via flow cytometry and fluorescence microscopy. Specifically designed to recognize activated caspase-3/7, these probes enable real-time monitoring of caspase-3/7 activity in living cells without suppressing or interfering with normal apoptotic progression. They label apoptotic cell nuclei with green fluorescence, which can be detected through the FITC channel by both fluorescence microscopy and flow cytometry. In addition, the staining procedure is compatible with subsequent cell fixation and permeabilization steps, supporting downstream multiplex immunofluorescence staining. According to experimental requirements, this reagent can be co-stained with Annexin V, PI, DAPI or other flow cytometry antibodies for combined apoptosis analysis (The Annexin V assay was applied to assess PS externalization in apoptotic cells).
Mechanistically, Caspase 3/7 Substrates (Green) integrate a caspase-3/7-specific recognition sequence (DEVD) with a high-affinity DNA-binding dye. Possessing excellent membrane permeability, the intact substrate readily crosses the plasma membrane into the cytoplasm. Unprocessed substrates exhibit no intrinsic fluorescence and display charge-based repulsion against genomic DNA. Upon apoptosis initiation, activated caspase-3/7 specifically cleaves the substrate, releasing the fluorescent DNA-binding moiety. The liberated dye subsequently binds tightly to nuclear DNA and emits intense green fluorescence, thereby accurately reflecting intracellular caspase-3/7 activity and visualizing characteristic apoptotic nuclear morphological alterations.
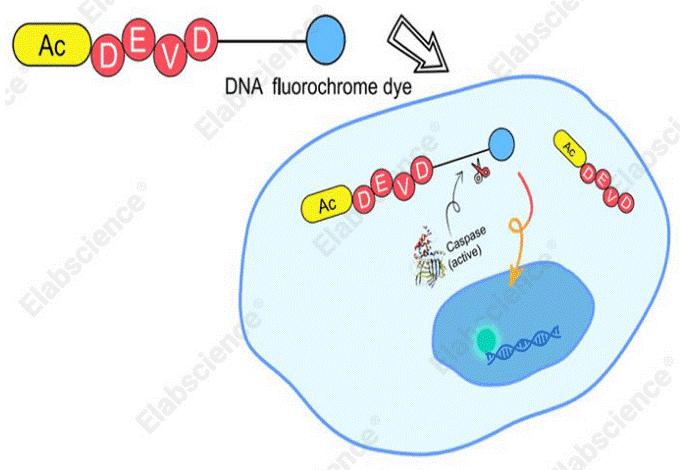
Fig. 5 Schematic diagram of the detection principle for caspase-3/7 activity in living cells.
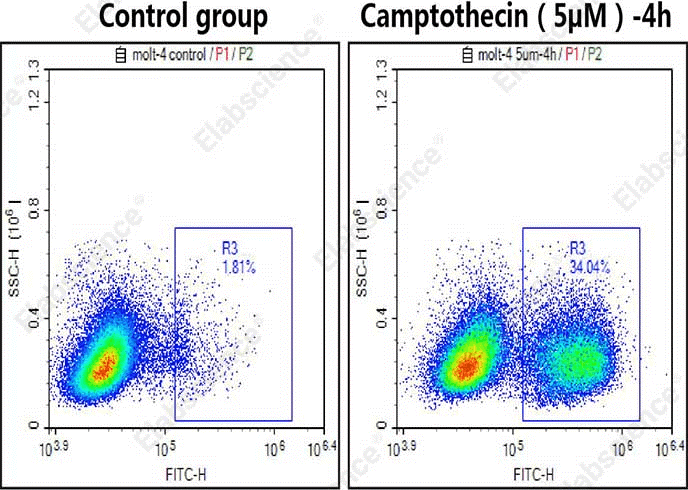
Fig. 6 Detection of caspase-3/7 activity in MOLT-4 cells. MOLT-4 cells were cultured with (left) or without (right) 5 μM camptothecin for 4 h and then stained with Caspase 3/7 Substrates (cat. no. E-CK-A483) and analyzed by flow cytometry. (The data are provided by Elabscience.)
05 Role of caspase-8 in extrinsic apoptosis and death receptor signaling
Caspase-8 serves as a core initiator caspase within death receptor-mediated extrinsic apoptosis, governing canonical programmed cell death signaling cascades. This pathway is triggered by extracellular death ligands that bind to matching death receptors on the cellular membrane. Representative ligands include FasL and TNFα, which respectively interact with Fas and TNFR1 receptors.
Following ligand-receptor binding, death receptors undergo trimerization and recruit the adaptor protein Fas-Associated Death Domain (FADD) to intracellular death domains. Consequently, these molecules assemble the Death-Inducing Signaling Complex (DISC), a pivotal upstream signaling platform. Via Death Effector Domain (DED) interactions, procaspase-8 monomers are concentrated at the DISC. Such induced proximity drives procaspase-8 dimerization and autocatalytic maturation, thereby generating enzymatically active caspase-8[12].
Notably, caspase-8 activation at the DISC represents a irreversible commitment step toward apoptotic cell fate (which can be analyzed by the caspase 8 activity assay). Once mature, active caspase-8 exerts multiple distinct biological functions.First, it directly processes downstream executioner caspases. In Type I cells, active caspase-8 immediately cleaves and activates procaspase-3 and procaspase-7. Conversion of these inactive zymogens enables widespread substrate proteolysis, which subsequently induces hallmark morphological alterations typical of apoptosis[13].
Second, caspase-8 amplifies apoptotic signaling through crosstalk with the mitochondrial intrinsic pathway in Type II cells. Specifically, active caspase-8 cleaves the BCL-2 family protein Bid (BH3-interacting domain death agonist) to produce truncated tBid. Subsequently, tBid translocates to mitochondria and induces mitochondrial outer membrane permeabilization (MOMP), promoting cytosolic release of cytochrome c and other pro-apoptotic factors. Cytochrome c further associates with Apaf-1 to assemble the apoptosome, which recruits and activates procaspase-9. Activated caspase-9 subsequently stimulates executioner caspases, thus establishing a sustained mitochondrial amplification loop that guarantees irreversible cell death even under weak death receptor stimulation[14].
In addition, caspase-8 negatively regulates necroptosis, an alternative programmed necrotic cell death modality. Mechanistically, caspase-8 cleaves and inactivates receptor-interacting protein kinase 1 (RIPK1) and RIPK3, core mediators of necroptotic signaling. Accordingly, caspase-8 deficiency or functional inhibition redirects death receptor-induced cell death from apoptosis toward necroptosis[15].
Tight spatiotemporal control of caspase-8 activation is essential to avoid excessive spontaneous cell death and maintain homeostasis under cellular stress. Caspase-8 is constitutively synthesized as an inactive precursor, and its maturation strictly depends on DISC assembly to support dimerization and auto-processing. Meanwhile, Inhibitors of Apoptosis Proteins (IAPs) also modulate apoptotic cascades, yet they predominantly target executioner caspases and caspase-9 rather than caspase-8[16].
Aberrant caspase-8 homeostasis closely correlates with diverse pathological disorders. Impaired caspase-8 activation, arising from mutations in death receptors or DISC-associated molecules, facilitates tumor immune escape and malignant progression. By contrast, uncontrolled excessive caspase-8 activity drives pathological excessive cell loss, which can be analyzed by the caspase 8 activity assay. Collectively, caspase-8 acts as a central signaling hub in extrinsic apoptosis, integrating extracellular death cues to precisely modulate orderly cellular apoptosis.
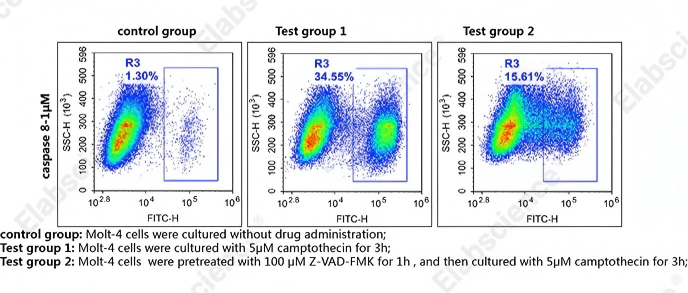
Fig. 7 Detection of caspase-8 activity in MOLT-4 cells. MOLT-4 cells were cultured with 5 μM camptothecin for 3 h (Test group 1), or pretreated with 100 μM Z-VAD-FMK for 1 h, then cultured with 5 μM camptothecin for 3 h (Test group 2), or without drug administration (control group). Cells were then stained with caspase-8 substrates (cat. no. E-CK-A488) and analyzed by flow cytometry. (The data are provided by Elabscience.)
06 Caspase-9 involvement in mitochondrial (intrinsic) apoptosis pathway
Caspase-9 acts as a central and indispensable initiator caspase in the intrinsic (mitochondrial) apoptotic pathway. This pathway is primarily triggered by diverse intracellular stresses, including DNA damage, growth factor deprivation, hypoxia, and endoplasmic reticulum stress. The commitment to apoptotic cell death via the intrinsic pathway is tightly regulated by mitochondrial outer membrane permeabilization (MOMP), a key event modulated by the Bcl-2 protein family[17].
Following MOMP, pro-apoptotic factors are released from the mitochondrial intermembrane space into the cytosol, with cytochrome c being the most prominent. Upon entering the cytosol, cytochrome c binds to Apoptotic protease activating factor-1 (Apaf-1), inducing a conformational change that drives Apaf-1 oligomerization. In the presence of ATP or dATP, this oligomerization leads to the assembly of a large heptameric protein complex, the apoptosome, which serves as a crucial activation platform for procaspase-9[18].
Procaspase-9, initially present as an inactive zymogen, is recruited to the apoptosome via homotypic interactions between its Caspase Recruitment Domain (CARD) and the CARD motifs of Apaf-1. The induced proximity of multiple procaspase-9 molecules within the apoptosome facilitates their dimerization and subsequent autocatalytic activation via proteolytic processing, thereby converting inactive procaspase-9 into active caspase-9[19].
Once activated, caspase-9 exerts its initiator caspase function by cleaving and activating downstream executioner caspases, primarily procaspase-3 and procaspase-7. This proteolytic cleavage removes the inhibitory pro-domains of executioner caspases, converting them into their active forms. These active executioner caspases then cleave a broad range of cellular substrates, mediating systematic cellular dismantling and inducing hallmark apoptotic morphological features, such as chromatin condensation, DNA fragmentation, and membrane blebbing[18,19].
The activation of caspase-9 within the apoptosome is tightly regulated, and its dysregulation has significant pathological implications, which can be analyzed by the caspase 9 activity assay. For example, insufficient activation of caspase-9 or other intrinsic pathway components can promote the survival of damaged cells, potentially contributing to tumorigenesis. Conversely, excessive or inappropriate caspase-9 activation leads to unintended cell death, which is implicated in neurodegenerative diseases and ischemic injury[19].
Although caspase-9’s primary role is confined to the intrinsic pathway, it engages in crosstalk with the extrinsic apoptotic pathway. In certain cell types (Type II cells), caspase-8, activated by death receptor signaling, cleaves the pro-apoptotic protein Bid into its truncated form (tBid). tBid then translocates to the mitochondria, promoting MOMP and subsequent cytochrome c release, thereby activating the intrinsic pathway and amplifying the apoptotic signal via caspase-9. This underscores the intricate regulatory network governing apoptosis, where caspase-9 activation by the apoptosome serves as a critical convergence point for intrinsic cell death signals.
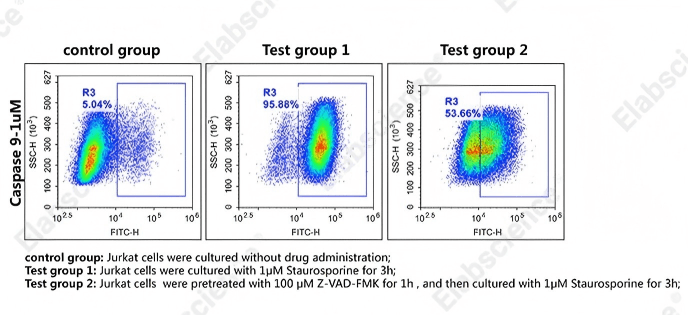
Fig. 8 Detection of caspase-9 activity in Jurkat cells. Jurkat cells were cultured with 1 μM camptothecin for 3 h (Test group 1), or pretreated with 100 μM Z-VAD-FMK for 1 h, then cultured with 1 μM camptothecin for 3 h (Test group 2), or without drug administration (control group). Cells were then stained with caspase-9 substrates (cat. no. E-CK-A489) and analyzed by flow cytometry. (The data are provided by Elabscience.)
References:
[1] Sahoo G, Samal D, Khandayataray P, et al. A review on caspases: key regulators of biological activities and apoptosis[J]. Molecular neurobiology, 2023, 60(10): 5805-5837.
[2] Chang H Y, Yang X. Proteases for cell suicide: functions and regulation of caspases[J]. Microbiology and molecular biology reviews, 2000, 64(4): 821-846.
[3] Chen M, Wang J. Initiator caspases in apoptosis signaling pathways[J]. Apoptosis, 2002, 7(4): 313-319.
[4] Sun G. Death and survival from executioner caspase activation[C]//Seminars in cell & developmental biology. Academic Press, 2024, 156: 66-73.
[5] Sansonetti P J, Phalipon A, Arondel J, et al. Caspase-1 activation of IL-1β and IL-18 are essential for Shigella flexneri–induced inflammation[J]. Immunity, 2000, 12(5): 581-590.
[6] Rathore R, McCallum J E, Varghese E, et al. Overcoming chemotherapy drug resistance by targeting inhibitors of apoptosis proteins (IAPs)[J]. Apoptosis, 2017, 22(7): 898-919.
[7] Yu H B, Finlay B B. The caspase-1 inflammasome: a pilot of innate immune responses[J]. Cell host & microbe, 2008, 4(3): 198-208.
[8] Creagh E M. Caspase crosstalk: integration of apoptotic and innate immune signalling pathways[J]. Trends in immunology, 2014, 35(12): 631-640.
[9] Jiang X. The intrinsic apoptotic pathway[J]. Cell Death: Mechanism and Disease, 2013: 15-40.
[10] Svandova E B, Veselá B, Lesot H, et al. Expression of Fas, FasL, caspase-8 and other factors of the extrinsic apoptotic pathway during the onset of interdigital tissue elimination[J]. Histochemistry and cell biology, 2017, 147(4): 497-510.
[11] Cenciarelli C. The role of mitotic spindle alterations in the induction of apoptosis[J]. 2009.
[12] Lüschen S, Falk M, Scherer G, et al. The Fas-associated death domain protein/caspase-8/c-FLIP signaling pathway is involved in TNF-induced activation of ERK[J]. Experimental cell research, 2005, 310(1): 33-42.
[13] Ikner A, Ashkenazi A. TWEAK induces apoptosis through a death-signaling complex comprising receptor-interacting protein 1 (RIP1), Fas-associated death domain (FADD), and caspase-8[J]. Journal of Biological Chemistry, 2011, 286(24): 21546-21554.
[14] Kantari C, Walczak H. Caspase-8 and bid: caught in the act between death receptors and mitochondria[J]. Biochimica et Biophysica Acta (BBA)-Molecular Cell Research, 2011, 1813(4): 558-563.
[15] Fritsch M, Günther S D, Schwarzer R, et al. Caspase-8 is the molecular switch for apoptosis, necroptosis and pyroptosis[J]. Nature, 2019, 575(7784): 683-687.
[16] Deveraux Q L, Roy N, Stennicke H R, et al. IAPs block apoptotic events induced by caspase‐8 and cytochrome c by direct inhibition of distinct caspases[J]. The EMBO journal, 1998, 17(8): 2215-2223.
[17] Kalkavan H, Green D R. MOMP, cell suicide as a BCL-2 family business[J]. Cell Death & Differentiation, 2018, 25(1): 46-55.
[18] Chong Z Z, Kang J Q, Maiese K. Apaf-1, Bcl-xL, cytochrome c, and caspase-9 form the critical elements for cerebral vascular protection by erythropoietin[J]. Journal of Cerebral Blood Flow & Metabolism, 2003, 23(3): 320-330.
[19] Marsden V S, O'Connor L, O'Reilly L A, et al. Apoptosis initiated by Bcl-2-regulated caspase activation independently of the cytochrome c/Apaf-1/caspase-9 apoptosome[J]. Nature, 2002, 419(6907): 634-637.



