The early detection of atherosclerotic plaques represents a critical window for intervention, in which natural killer (NK) cells and macrophages play fundamentally distinct yet interconnected roles.
This review systematically examines the involvement of NK cell cytotoxicity in atherosclerotic lesion initiation and addresses whether NK cells infiltrate plaques earlier than macrophages. We further discuss the divergent functions of CD68+ macrophages and NKG2D+ NK cells within the plaque microenvironment, as well as the impact of macrophage polarization on the precision of early plaque detection. Furthermore, we evaluate the diagnostic value of combining NK cell and macrophage markers to improve early plaque identification, and compare these immune cells as potential biomarkers in the setting of metabolic syndrome.
Table of Contents
1. What is the role of NK cell cytotoxicity in early lesion development?
2. Do NK cells infiltrate atherosclerotic plaques earlier than macrophages?
3. Roles of CD68+ macrophages and NKG2D+ NK cells in plaque microenvironment
4. Macrophage polarization influences early plaque detection accuracy
5. Combining NK and macrophage markers enhances early plaque diagnosis
01 What is the role of NK cell cytotoxicity in early lesion development?
The role of natural killer (NK) cell cytotoxicity in early atherosclerotic lesion development remains intensively investigated. Recent high-impact studies have concurred that NK cell-mediated cytotoxicity is not a primary driver of initial plaque formation. Although NK cells exert potent cytotoxicity via perforin/granzyme pathways and death receptor signaling, these mechanisms target virally infected or transformed cells, and their contribution to early atherosclerosis is minimal. This conclusion is strongly supported by rigorous genetic and functional studies in murine models, which overcome the cellular specificity limitations of earlier pharmacological depletion approaches.
A pivotal study using precise genetic tools to deplete NK cells or render them hyperresponsive showed no significant alterations in atherosclerotic lesion size or composition in standard murine models (e.g., Ldlr-/- mice fed a high-fat diet)[1]. This finding challenges the hypothesis that NK cell cytotoxicity against stressed endothelial cells or nascent foam cells initiates lesion formation[1]. Critical to this study was the use of Ncr1iCre mice, which specifically target NK cells without affecting other lymphocyte populations, enabling unprecedented isolation of NK cell function[2]. The absence of phenotypic changes in atherosclerosis with both loss and gain of NK cell function confirms that their cytotoxic potential is dispensable for early lesion development.
Comprehensive meta-analyses of the immune landscape within atherosclerotic plaques further corroborate this conclusion. Integration of single-cell RNA sequencing and mass cytometry data from multiple independent studies has confirmed that NK cells constitute a minor aortic infiltrate in diseased mice, in contrast to the dominant myeloid lineages (particularly macrophages)[1,2].
Furthermore, human clinical data demonstrate that the frequency of CD56bright NK cells, a subset more specialized in cytokine production than cytotoxicity, is positively correlated with increased atheroburden and features of plaque instability. For example, their interferon-gamma (IFN-γ) production potently activates macrophages, thereby exacerbating plaque inflammation and enhancing vulnerability to rupture in advanced disease stages[2,3].
The current scientific consensus defines a clear temporal and functional distinction between these cell populations. Compared with macrophages, NK cells act as secondary contributors, with their primary impact likely occurring in advanced disease stages, where IFN-γ and other inflammatory mediators regulate plaque inflammation and stability. Although NK cells are integral components of the complex immune network within atherosclerotic plaques, macrophages are unequivocally the primary effectors of disease initiation[3].
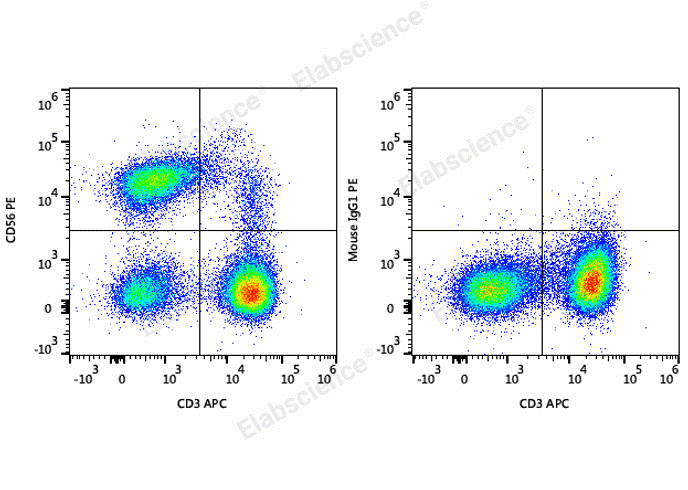
Fig. 1 Detection of NK cells in human peripheral blood lymphocytes. Human peripheral blood lymphocytes are stained with APC Anti-Human CD3 Antibody and PE Anti-Human CD56 Antibody (Left). Lymphocytes are stained with APC Anti-Human CD3 Antibody and PE Mouse IgG1, κ Isotype Control (Right). (The datas are provided by Elabscience.)
_.png)
Fig. 2 Detection of TBNK cell subsets in human peripheral blood mononuclear cells (PBMCs). PBMCs were stained with Anti-Human CD19-FITC/CD56-PE/CD3-PE/Cyanine7/CD45-PerCP Cocktail (E-AB-FC0011), and subsequently analyzed by flow cytometry. (The datas are provided by Elabscience.)
Elabscience® Quick Overview of Popular Products:
Table 1. Reagents for NK cells Research
|
Product Name |
Cat. No. |
|
PE Anti-Human CD56/NCAM Antibody[5.1H11] |
E-AB-F1239D |
|
PE Mouse IgG1, κ Isotype Control[MOPC-21] |
E-AB-F09792D |
|
APC Anti-Human CD3 Antibody[OKT-3] |
E-AB-F1001E |
|
EasySort™ Mouse NK Cell Isolation Kit |
MIM005N |
|
EasyStain™ Human Fc Receptor Blocking Solution |
E-CK-A171 |
|
STYX™ Violet Fixable Viability Kit |
E-CK-A167 |
|
STYX™ Near-IR Fixable Viability Kit |
E-CK-A168 |
|
STYX™ Green Fixable Viability Kit |
E-CK-A166 |
|
STYX™ Red Fixable Viability Kit |
E-CK-A170 |
|
STYX™ Yellow Fixable Viability Kit |
E-CK-A169 |
|
Anti-Human CD45-FITC/CD3-PE/Cyanine5/CD56/NCAM-PE/CD19-PE/Elab Fluor® 594 Cocktail |
E-AB-FC0028 |
|
Anti-Human CD3-FITC/CD16+CD56-PE Cocktail |
E-AB-FC0001 |
|
Anti-Human CD19-FITC/CD56-PE/CD3-PE/Cyanine7/CD45-PerCP Cocktail |
E-AB-FC0011 |
02 Do NK cells infiltrate atherosclerotic plaques earlier than macrophages?
Natural killer (NK) cells are not early infiltrators in atherosclerotic plaques. Macrophages represent one of the earliest and most abundant immune cell populations recruited to nascent atherosclerotic lesions, whereas NK cell infiltration occurs at later stages and is quantitatively negligible.
In line with this notion, NK cells do not contribute to the initial wave of immune infiltration. Comprehensive analyses of human atherosclerotic plaques across disease stages consistently identify macrophages as the predominant innate immune cell population, with substantial frequencies of T lymphocytes and dendritic cells, whereas NK cells are detectable only at extremely low levels. A meta‑analysis of leukocyte heterogeneity in atherosclerotic mouse aortas further corroborates these observations, confirming diverse macrophage subsets and identifying NK cells as a minor component of the inflammatory infiltrate[1,2,3].
Although NK cells are present in established human plaques and their peripheral frequencies correlate with elevated atheroburden and plaque instability, their functional role is regarded as modulatory rather than fundamental. Consistent with this, studies using selective NK cell depletion or hyperactivation in genetically modified mouse models show no significant effects on atherosclerotic lesion development, indicating that NK cells are not indispensable drivers of core pathogenic processes. Instead, NK cells likely act during later plaque progression by secreting pro‑inflammatory cytokines such as IFN‑γ and exerting cytotoxicity against stressed vascular cells, thereby modulating plaque stability. Importantly, these NK cell‑mediated functions depend on a plaque microenvironment already established by macrophages and other inflammatory cells[2,3,4].
Thus, the temporal dynamics of immune cell infiltration firmly establish macrophages as pioneering innate immune sentinels in early atherogenesis, whereas NK cells play a secondary role that manifests only after the plaque inflammatory framework has been established.
03 Roles of CD68+ macrophages and NKG2D+ NK cells in plaque microenvironment
The roles of CD68+ macrophages and NKG2D+ natural killer (NK) cells within the atherosclerotic plaque microenvironment represent two distinct yet interconnected facets of vascular inflammation, with macrophages serving as central structural and functional architects of the lesion, while NK cells, particularly those expressing the activating receptor NKG2D, act as modulatory effectors whose influence is more pronounced in later stages of disease progression[2,3,4].
These CD68+ macrophages are not merely passive lipid scavengers, they are dynamic regulators of plaque biology. Beyond lipid accumulation, CD68+ macrophages secrete a vast array of pro-inflammatory cytokines (e.g., TNF-α, IL-1β, IL-6), matrix metalloproteinases (MMPs) that degrade the fibrous cap, and reactive oxygen species that perpetuate endothelial dysfunction and oxidative stress[2,3,4].
NKG2D+ NK cells are activated by stress-induced ligands on damaged vascular cells and are present at low frequencies in human atherosclerotic plaques. Circulating CD56bright NKG2D+ NK cells correlate with increased atheroburden and plaque instability. These cells exert cytotoxicity and secrete IFN‑γ, which promotes pro‑inflammatory macrophage polarization and plaque vulnerability. However, mouse studies reveal that NK cell depletion or hyperactivation does not affect early atherosclerotic lesion development. Thus, NKG2D+ NK cells contribute to inflammatory progression in advanced plaques but are not essential initiators of atherogenesis[2,3,4].
The interplay between macrophages and NK cells in atherosclerosis is hierarchical and stage-dependent. Macrophages shape the foundational inflammatory plaque microenvironment from early stages, thereby inducing NKG2D ligand expression on vascular cells and enabling subsequent NK cell recruitment and activation. NKG2D+ NK cells act as modulators rather than initiators, promoting inflammation and plaque vulnerability in advanced lesions primarily via IFN‑γ secretion. As context-dependent amplifiers of plaque inflammation rather than essential drivers of atherogenesis, NK cells represent a viable target for stabilizing high-risk plaques, whereas macrophages remain the priority for preventing early lesion formation[2,3,4].
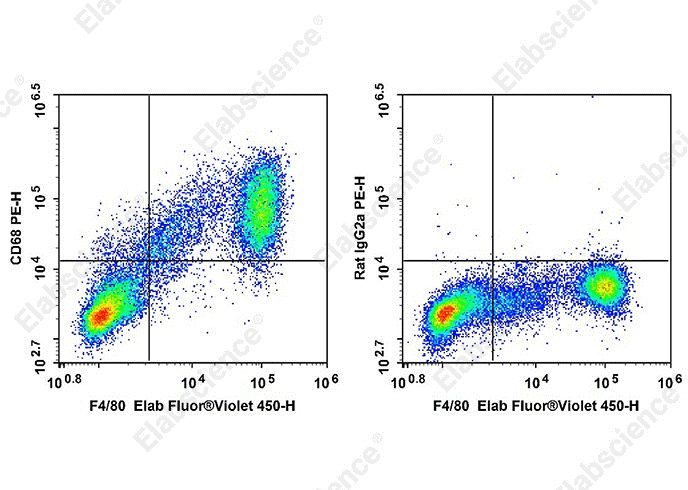
Fig. 3 Detection of C57BL/6 murine abdominal macrophages. Staining of C57BL/6 murine abdominal macrophages elicited by Thioglycolate are stained with Elab Fluor® Violet 450 Anti-Mouse F4/80 Antibody[CI:A3-1] and intracellular stained with PE Anti-Mouse CD68 Antibody[FA-11] (left) or PE Rat IgG2a Isotype Control (right). (The datas are provided by Elabscience.)
Elabscience® Quick Overview of Popular Products:
Table 2. Reagents for Macrophages Research
|
Product Name |
Cat. No. |
|
PE Anti-Mouse CD68 Antibody[FA-11] |
AN00927D |
|
PE Rat IgG2a, κ Isotype Control[2A3] |
E-AB-F09832D |
|
Elab Fluor® Violet 450 Anti-Mouse F4/80 Antibody[CI:A3-1] |
E-AB-F0995Q |
|
RAW 264.7 Polarized M1 Macrophage Induction and Identification Kit |
XJM004 |
|
APC Anti-Mouse CD163 Antibody[S15049F] |
E-AB-F1295E |
|
PE Anti-Mouse CD86 Antibody[GL-1] |
E-AB-F0994D |
|
APC Anti-Mouse CD8 Antibody[YTS-169] |
AN00576E |
|
Elab Fluor® Violet 450 Anti-Mouse CD8a Antibody[53-6.7] |
E-AB-F1104Q |
|
Caspase 1 Activity Assay Kit(Colorimetric Method) |
E-CK-A381 |
|
Caspase 1 Activity Detection Substrate for Flow Cytometry |
E-CK-A481 |
|
Reactive Oxygen Species (ROS) Fluorometric Assay Kit (Red) |
E-BC-F005 |
|
Lactate Dehydrogenase (LDH) Cytotoxicity Colorimetric Assay Kit |
E-BC-K771-M |
|
PE Anti-Mouse IL-6 Antibody[MP5-20F3] |
E-AB-F1207UD |
|
FITC Anti-Human IL-6 Antibody[MQ2-13A5] |
E-AB-F1206C |
|
Aptplex™ Human Immunosuppression 5-Plex Panel |
MPA002 |
|
Human IL-6 (Interleukin 6) ELISPOT Kit |
ESP-H0009 |
|
High Sensitivity Mouse IL-6 (Interleukin 6) ELISA Kit |
E-HSEL-M0003 |
|
FITC Anti-Human IL-1 beta Antibody[CRM56] |
AN00842C |
|
Human IL-1β(Interleukin 1 Beta) ELISA Kit |
E-EL-H0149 |
|
Mouse IL-1β(Interleukin 1 Beta) ELISA Kit |
E-EL-M0037 |
|
Rat IL-1β(Interleukin 1 Beta) ELISA Kit |
E-EL-R0012 |
04 Macrophage polarization influences early plaque detection accuracy
Macrophage polarization is a critical determinant governing the accuracy of early atherosclerotic plaque detection, representing an important interface between cardiovascular immunology and diagnostic imaging. As the predominant immune cells in early atherosclerotic lesions, macrophages exhibit remarkable phenotypic plasticity, ranging from pro-inflammatory M1-like macrophages (M1 macrophages) characterized by high expression of CD80, CD86, and MHC-II, to anti-inflammatory and reparative M2-like subtypes (M2 macrophages) marked by CD206, CD163, and arginase-1. Advanced single-cell profiling has further identified functionally distinct subpopulations, including Trem2+ foamy macrophages associated with lipid metabolism and efferocytosis, as well as interferon-inducible cells (IFNICs) (e.g., THP-1 macrophages, Raw 264.7 macrophages) with strong pro-inflammatory transcriptional signatures. These subsets differ dramatically in metabolic activity, cytokine secretion, and lipid handling, all of which shape the biochemical and structural features of nascent plaques[5,6].
Conventional imaging modalities such as 18F-fluorodeoxyglucose positron emission tomography (18F-FDG PET) primarily detect the elevated glycolytic metabolism characteristic of M1-polarized macrophages, and therefore preferentially identify inflamed M1-rich plaques while underestimating lesions dominated by M2, Trem2+, or other metabolically quiescent subsets, creating diagnostic blind spots and false negatives. Furthermore, the temporal differentiation and spatial heterogeneity of macrophage polarization within plaques further reduce the reliability of single-timepoint imaging. Although emerging polarization-specific probes targeting CD206, folate receptor β, or other subset-restricted markers show potential for improved discrimination, most remain preclinical. Current clinical imaging strategies fail to fully capture macrophage phenotypic diversity, often leading to inaccurate risk stratification. Thus, next-generation imaging approaches incorporating multiplexed tracers or machine learning-assisted signal interpretation are essential to account for macrophage heterogeneity and enhance the precision of early atherosclerosis detection[7].
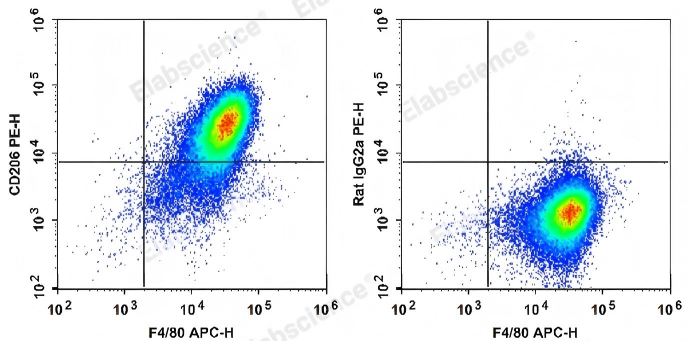
Fig. 4 Detection of C57BL/6 murine abdominal macrophages. C57BL/6 murine abdominal macrophages elicited by starch broth are stained with APC Anti-Mouse F4/80 Antibody and PE Anti-Mouse CD206 Antibody (Left). Abdominal macrophages are stained with APC Anti-Mouse F4/80 Antibody and PE Rat IgG2a, κ Isotype Control (Right). (The datas are provided by Elabscience.)
Elabscience® Quick Overview of Popular Products:
Table 3. Reagents for Mouse Macrophages Research
|
Product Name |
Cat. No. |
|
PE Anti-Mouse CD206/MMR Antibody[C068C2] |
E-AB-F1135D |
|
PE Rat IgG2a, κ Isotype Control[2A3] |
E-AB-F09832D |
|
APC Anti-Mouse F4/80 Antibody[CI:A3-1] |
E-AB-F0995E |
|
FITC Anti-Mouse/Human CD11b Antibody[M1/70] |
E-AB-F1081C |
|
Elab Bright™ Violet 421 Anti-Mouse CD86 Antibody[GL-1] |
E-AB-F0994Q2 |
|
STYX™ Violet Fixable Viability Kit |
E-CK-A167 |
|
STYX™ Near-IR Fixable Viability Kit |
E-CK-A168 |
05 Combining NK and macrophage markers enhances early plaque diagnosis
Atherosclerosis is a chronic inflammatory disorder characterized by lipid accumulation and immune cell infiltration in arterial walls. Vulnerable plaques, which are prone to rupture, pose a high risk of acute cardiovascular events such as myocardial infarction and stroke. Thus, their early identification is pivotal for effective clinical intervention and prevention of adverse outcomes[8,9,10].
The combined analysis of natural killer (NK) cell and macrophage markers significantly improves the early diagnosis and characterization of vulnerable atherosclerotic plaques. This approach enhances diagnostic sensitivity and specificity by capturing the heterogeneity and activation states of immune cells within the plaque microenvironment, addressing a critical unmet need in cardiovascular disease management[11].
Macrophages are central to the initiation and progression of atherosclerosis, displaying substantial phenotypic plasticity that is tightly regulated by local microenvironmental cues[12]. These cells infiltrate lesions at the early stages of atherogenesis, exerting dual roles: beneficial functions include the clearance of cytotoxic lipoproteins and apoptotic cells, while detrimental effects involve promoting inflammation and exacerbating plaque instability. Distinct macrophage subsets, identifiable by specific phenotypic markers (e.g., CD68 as a pan-macrophage marker, CD163 as a marker for anti-inflammatory M2-like macrophages), are associated with different stages and pathological characteristics of atherosclerotic plaques[13]. Furthermore, studies integrating single-cell RNA sequencing (scRNA-seq) have uncovered novel foamy macrophage subpopulations in human atherosclerotic plaques, highlighting the remarkable cellular complexity and heterogeneity of macrophages within lesion microenvironments[14].
As mentioned above, NK cells also contribute to atherosclerotic progression, particularly in the regulation of plaque vulnerability. Emerging evidence indicates that unstable human coronary artery plaques are associated with increased frequencies of circulating CD56bright NK cells[15]. CD56 serves as a canonical surface marker for NK cells, and the CD56bright subset is well-characterized by its potent immunomodulatory capabilities, including robust cytokine secretion. Notably, infiltration of CD56bright NK cells has also been observed in symptomatic carotid atherosclerotic plaques, suggesting that NK cells actively participate in shaping the inflammatory microenvironment of plaques and thereby modulate their stability[15,16].
The integration of NK cell and macrophage markers enables a more nuanced and comprehensive approach to plaque characterization. For instance, the co-expression of macrophage markers (e.g., CD68 for pan-macrophage identification, CD163 for M2-like subset distinction) and NK cell markers (e.g., CD56 for NK cell enumeration) provides a holistic view of the immune cell landscape within atherosclerotic plaques. This dual-marker strategy can reveal synergistic immune cell infiltration patterns and crosstalk, such as NK cell-macrophage interactions mediated by interferon-gamma (IFN-γ) and tumor necrosis factor-alpha (TNF-α), which may precede structural plaque changes detectable by conventional imaging modalities. Importantly, the inflammatory microenvironment driven by these cellular interactions is a key determinant of atherosclerotic plaque erosion and rupture, which underscores the clinical value of combined marker analysis[17,18].
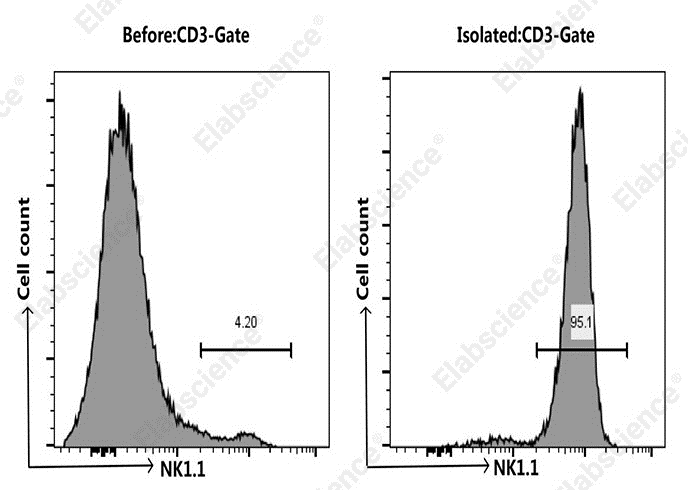
Fig. 5 Analysis of NK cells in spleen cells from C57BL/6 murine before and after sorting. NK cells were isolated from the spleen cells of C57BL/6 mice, mouse spleen cells were co-stained before and after the sorting with APC Anti-Mouse CD3 Antibody[17A2] (E-AB-F1013E) and PE Anti-Mouse NK1.1 Antibody[PK136] (E-AB-F0987D), the result shows that the proportion of CD3-NK1.1+ cells before and after the sorting of mouse spleen cells were 4.2% and 95.1%, respectively. (The datas are provided by Elabscience.)
References:
[1] Zernecke A, Winkels H, Cochain C, et al. Meta-analysis of leukocyte diversity in atherosclerotic mouse aortas[J]. Circulation research, 2020, 127(3): 402-426.
[2] Nour-Eldine W, Joffre J, Zibara K, et al. Genetic depletion or hyperresponsiveness of natural killer cells do not affect atherosclerosis development[J]. Circulation research, 2018, 122(1): 47-57.
[3] Kothari H, Drago F, McSkimming C, et al. Peripheral CD56bright Natural Killer Cells Associate With Increased Atheroburden and Unstable Plaque Features in Humans[J]. Circulation, 2020, 142(Suppl_3): A15301-A15301.
[4] Palano M T, Cucchiara M, Gallazzi M, et al. When a friend becomes your enemy: natural killer cells in atherosclerosis and atherosclerosis-associated risk factors[J]. Frontiers in immunology, 2022, 12: 798155.
[5] Libby P. Inflammation and the pathogenesis of atherosclerosis[J]. Vascular Pharmacology, 2024, 154: 107255.
[6] Tearney G J, Yabushita H, Houser S L, et al. Quantification of macrophage content in atherosclerotic plaques by optical coherence tomography[J]. Circulation, 2003, 107(1): 113-119.
[7] Patterson M T, Firulyova M M, Xu Y, et al. Trem2 promotes foamy macrophage lipid uptake and survival in atherosclerosis[J]. Nature cardiovascular research, 2023, 2(11): 1015-1031.
[8] Schneider M K, Wang J, Kare A, et al. Combined near infrared photoacoustic imaging and ultrasound detects vulnerable atherosclerotic plaque[J]. Biomaterials, 2023, 302: 122314.
[9] Zhang X, Centurion F, Misra A, et al. Molecularly targeted nanomedicine enabled by inorganic nanoparticles for atherosclerosis diagnosis and treatment[J]. Advanced drug delivery reviews, 2023, 194: 114709.
[10] Gaba P, Gersh B J, Muller J, et al. Evolving concepts of the vulnerable atherosclerotic plaque and the vulnerable patient: implications for patient care and future research[J]. Nature Reviews Cardiology, 2023, 20(3): 181-196.
[11] Wang J, Kang Z, Liu Y, et al. Identification of immune cell infiltration and diagnostic biomarkers in unstable atherosclerotic plaques by integrated bioinformatics analysis and machine learning[J]. Frontiers in Immunology, 2022, 13: 956078.
[12] Goossens P, Lu C, Cao J, et al. Integrating multiplex immunofluorescent and mass spectrometry imaging to map myeloid heterogeneity in its metabolic and cellular context[J]. Cell metabolism, 2022, 34(8): 1214-1225. e6.
[13] Lavie L, Si-On E, Hoffman A. Giant phagocytes (Gφ) and neutrophil-macrophage hybrids in human carotid atherosclerotic plaques–An activated phenotype[J]. Frontiers in Immunology, 2023, 14: 1101569.
[14] Lu Y, Wu S, Zhu S, et al. Integrated single-cell analysis revealed novel subpopulations of foamy macrophages in human atherosclerotic plaques[J]. Biomolecules, 2024, 14(12): 1606.
[15] Hunter-Chang M, Flora C L, Kothari H, et al. Unstable coronary artery plaque features in humans are associated with higher frequency of circulating CD56bright natural killer cells[J]. Journal of Vascular Research, 2025, 62(4): 204-210.
[16] Ishwar D, Haldavnekar R, Venkatakrishnan K, et al. Minimally invasive detection of cancer using metabolic changes in tumor-associated natural killer cells with Oncoimmune probes[J]. Nature communications, 2022, 13(1): 4527.
[17] Ge P, Li H, Ya X, et al. Single-cell atlas reveals different immune environments between stable and vulnerable atherosclerotic plaques[J]. Frontiers in Immunology, 2023, 13: 1085468.
[18] Shen L, Chen M, Su Y, et al. NIR-II imaging for tracking the spatiotemporal immune microenvironment in atherosclerotic plaques[J]. ACS nano, 2024, 18(50): 34171-34185.



