In flow cytometry experiments, implementing Fc receptor blocking is a crucial step to ensure data accuracy, especially when analyzing myeloid cell surface markers. In this post, we will introduce the necessity of Fc receptor blocking in flow cytometry detection and analyze the consequences that may arise if blocking is not performed.
Table of Contents
1. Why is Fc Blocking Necessary in Flow Cytometry?
2. Potential Consequences of Not Performing Fc Blocking
3. Conventional Fc Blocking Methods
4. Establishing a Blocking Efficacy Evaluation System
5. Fc Receptor Blocking Experimental Procedure
01 Why is Fc Blocking Necessary in Flow Cytometry?
Antibodies commonly used in flow cytometry are composed of two functional regions: the Fab segment and the Fc segment. The Fab segment is responsible for specifically recognizing antigens, while the Fc segment is the key part of the antibody that mediates effector functions. Various immune cells in the human body express different classes of Fc receptors on their surfaces, including Fc receptors for IgG (FcγR), IgE (FcεR), IgA (FcαR), IgM (FcμR), and IgD (FcδR). Among these, IgG is the most common antibody type, making Fcγ receptors, which can bind to IgG, particularly important.
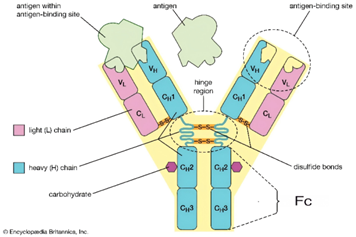
Fig. 1 Schematic Diagram of Antibody Structure.
Fcγ receptors are primarily divided into six subtypes: FcγRI (CD64), FcγRIIa (CD32a), FcγRIIb (CD32b), FcγRIIc (CD32c), FcγRIIIa (CD16a), and FcγRIIIb (CD16b). Under physiological conditions, these receptors mediate critical immune effector functions. However, in flow cytometry assays, they are prone to causing non-specific binding.
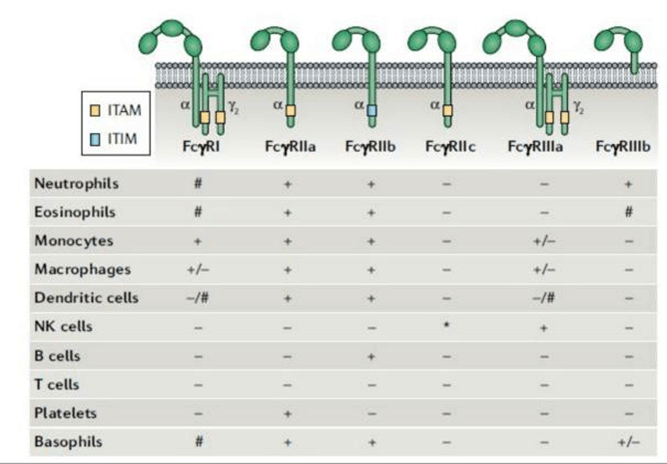
Fig. 2 Major Types of Fcγ Receptors and Their Distribution.
During antibody incubation, the Fc segment can bind non-specifically to Fc receptors on the cell surface, generating fluorescent signals unrelated to the target antigen. This non-specific binding exhibits the same signal characteristics as specific binding in flow cytometry, making it difficult to distinguish through routine data analysis and leading to biased results.
02 Potential Consequences of Not Performing Fc Blocking
Failure to perform Fc blocking may lead to the following issues:
2.1 Elevated background fluorescence and reduced signal-to-noise ratio
Unblocked Fc receptors can bind to the Fc region of flow cytometry antibodies, resulting in significant non-specific fluorescent signals and increased background noise. This significantly reduces the signal-to-noise ratio, particularly when detecting low-expression antigens, where the target signal may be overwhelmed by the background, leading to false-negative results.
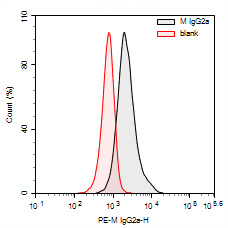
Fig. 3 Flow Cytometry Results of THP-1 Cells: Comparison between PE Mouse IgG2a Isotype Control Antibody (Host: Mouse) and the Unstained Control Group (Blank).
2.2 False-positive signals interfere with experimental results
The expression of Fc receptors is regulated by the activation state of cells and cytokines. Pro-inflammatory cytokines (such as IL-6 and TNF-α) typically promote the expression of activating Fc receptors, while anti-inflammatory cytokines enhance the expression of inhibitory receptors. Specifically, under certain inflammatory conditions, myeloid cells (e.g., monocytes/macrophages) upregulate the expression of Fc receptors such as FcεRIα, FcγRIIA, and FcγRIIB on their surface after activation. This increases the likelihood of non-specific binding by Fc receptors, which may lead to false-positive results.
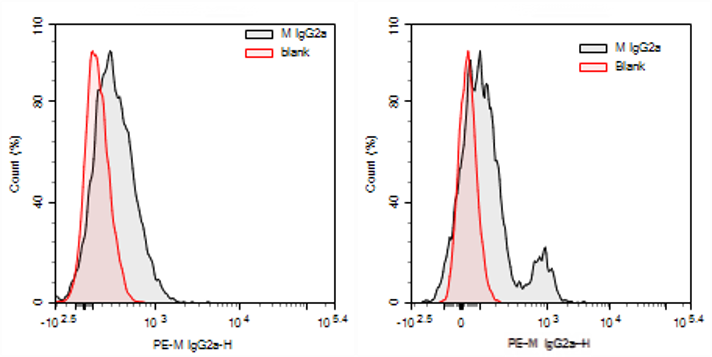
Fig. 4 Detection Results of Human Peripheral Blood PBMCs. (Left: Staining results of resting human peripheral blood PBMCs with PE Mouse IgG2a isotype antibody versus unstained Blank group; Right: Staining results of activated human peripheral blood PBMCs with PE Mouse IgG2a isotype antibody versus unstained Blank group).
03 Conventional Fc Blocking Methods
Conventional Fc blocking methods are primarily divided into serum blocking and specific antibody blocking.
3.1 Serum Blocking Method
Uses excess non-specific immunoglobulins from the same species (e.g., fetal bovine serum or human serum) to competitively bind Fc receptors on the cell surface. Its advantages include lower cost; however, drawbacks include potential non-specific background interference from other protein components in the serum, lower blocking efficiency compared to specific antibody methods, and poor effectiveness for cells with high Fc receptor expression.
3.2 Specific Antibody Blocking Method
Employs monoclonal antibodies against CD16, CD32, CD64, and other blocking reagents to directly prevent the binding of Fcγ receptors to the Fc segment of antibodies. These blocking agents are designed for ready-to-use convenience, streamlining the experimental process and offering higher blocking efficiency. This method is particularly suitable for cells expressing high levels of Fc receptors or experiments requiring high detection sensitivity, such as flow cytometry for immune cells.
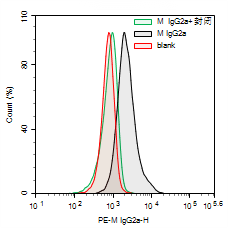
Fig. 5 Experimental Results of THP-1 Cell Detection. (Red represents the unstained Blank group, black represents the PE Mouse IgG2a isotype antibody staining group, and green represents the PE Mouse IgG2a isotype antibody staining group with Human Fc Receptor Blocking Solution blocking).
04 Establishing a Blocking Efficacy Evaluation System
Establishing an effective blocking efficacy evaluation system is a critical step in ensuring the reliability of experimental data. This involves not only the use of blocking solutions prior to experiments but also the validation of whether blocking has genuinely taken effect post-experiment through appropriate controls and data analysis.
Table 1. Reference for Establishing an Fc Receptor Blocking Efficacy Evaluation System
|
Control Group |
Purpose |
Blocking Efficacy Evaluation |
|
Unstained Cells |
Define background fluorescence Used to adjust the detection voltage of the flow cytometer and determine the level of cellular autofluorescence and noise. Serves as the baseline for all negative signals. |
It is not directly used to assess blocking efficacy but serves as a comparative baseline for other controls. |
|
Unblocked Isotype Control |
Defining the "baseline" of non-specific background It simulates the non-specific binding between the antibody Fc region and Fc receptors. A higher signal indicates more severe background binding mediated by the cell's own Fc receptors. |
Compared to the "Blocked Isotype Control," the signal in this group may be significantly higher. (In some samples, the absence of blocking may have limited impact on the results.) |
|
Blocked Isotype Control |
Assess blocking efficacy This is the most critical group. It shows the remaining level of Fc receptor non-specific binding after treatment with a blocking reagent. |
Success Criterion: Its fluorescence intensity (MFI) is significantly reduced and is very close to that of the "unstained cells" group. Quantitative Criterion: Blocking efficiency = [1 - (MFI of this group / MFI of the unblocked isotype control)] × 100% > 80%. |
|
Blocked Specific Antibody |
Confirm the true signal under effective blocking conditions Under ideal background conditions, it displays the true expression level of the target antigen. Used to calculate the final signal-to-noise ratio. |
Compared with the "Blocked Isotype Control," the specific signal should be clearly distinguishable with a high signal-to-noise ratio. Success Criterion: Signal-to-Noise Ratio = (MFI of this group / MFI of the Blocked Isotype Control). This value should be as large as possible. |
05 Fc Receptor Blocking Experimental Procedure
Taking EasyStain™ Human Fc Receptor Blocking Solution as an example, the standardized blocking procedure is as follows:
(1) Prepare a single-cell suspension according to the experimental requirements, ensuring that cell viability and single-cell rate meet the standards. Resuspend the cells in cell staining buffer and adjust the cell density to 5-10 × 106 cells/mL.
(2) Take 100 μL of the cell suspension (containing 0.5-1 million cells) and add it to a test tube. This constitutes one reaction system. Aliquot according to the number of experimental samples (including experimental and control groups).
(3) Precisely add 1 Test (5 μL) of EasyStain™ Human Fc Receptor Blocking Solution to each reaction system containing 100 μL of cell suspension.
(4) Mix gently by pipetting or vortex briefly. Then, incubate in the dark at room temperature (18-25°C) for 10 minutes.
(5) After incubation, there is no need to wash the cells. Proceed directly with fluorescently labeled antibody staining according to the corresponding antibody protocol.
In flow cytometry experiments, Fc receptor blocking plays a crucial role. It helps prevent the impact of non-specific binding mediated by Fc receptors on experimental results. Choosing an appropriate blocking strategy not only enhances experimental efficiency but also ensures the scientific validity and credibility of research conclusions.



