Phosphorylation as an effective way to regulate proteins is responsible for many intracellular processes such as cell growth and development, signal transduction and metabolism. Any disorders in the phosphorylation process are likely to drive the inappropriate activation or deactivation of the cellular processes, resulting in the break of the delicate balance in the cells. It is believed that many human diseases including different cancers have been associated with the abnormal phosphorylation of cellular proteins.
Phosphorylation of a protein is characterized by a reversible, enzymatically mediated mechanism. Upon reception of a signal, a protein is phosphorylated by the addition of a covalently bound phosphate group to its amino acid side chains in a few seconds.
In the process of phosphorylation, a phosphate group from adenosine triphosphate (ATP) can be transferred to the amino acid residue of a protein under the control of protein kinases, while phosphatases can remove the phosphate group from the protein.
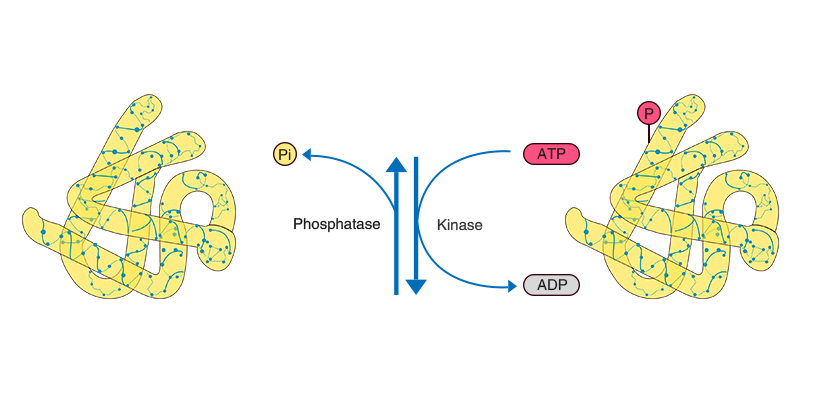
One-third of the proteins present in a mammalian cell are phosphorylated at one time or another. There is more than one phosphorylation site in half of these proteins, and different sites are able to elicit different cellular responses. The most common amino acids to be phosphorylated in eukaryotic are serine, threonine, and tyrosine because of their hydroxyl groups (-OH).
Elabscience has a wide range of ELISA kits and antibodies for the study associated with protein phosphorylation. Phosphatase inhibitors are indispensable in the study of protein phosphorylation when you prepare samples for the experiments such as western blotting analysis. Discover Elabscience's resources to support your related research.



