CONTENTS
■ BIOMEDICAL IMPORTANCE OF PROTEIN
■ STRUCTURAL ORGANIZATION OF PROTEINS
1. DEFINITION
Proteins are the high molecular weight material which on complete hydrolysis gives amino acids. The word protein is derived from Greek word Proteios means primary or holding first place or pre-eminent [1].
2. BIOMEDICAL IMPORTANCE OF PROTEIN
They are:
1). Proteins are the main structural components of the cytoskeleton.
2). They are the sole source to replace Nitrogen of the body.
3). Biochemical catalysts known as enzymes are proteins.
4). Proteins known as immunoglobulins serve as the first line of defence against bacterial and viral infections.
5). Several hormones are protein in nature.
6). Structural proteins furnish mechanical support and some of them like actin and myosin are contractile proteins and help in the movement of muscle fibre, microvilli etc.
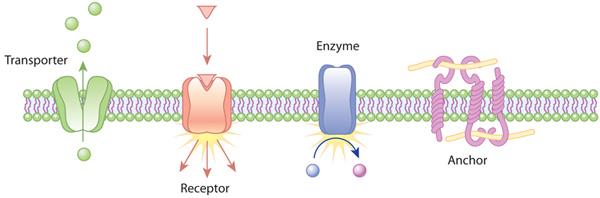
7). Some proteins present in cell membrane, cytoplasm and nucleus of the cell act as receptors.
8). The transport proteins carry out the function of transporting specific substances either across the membrane or in the body fluids [1&2].
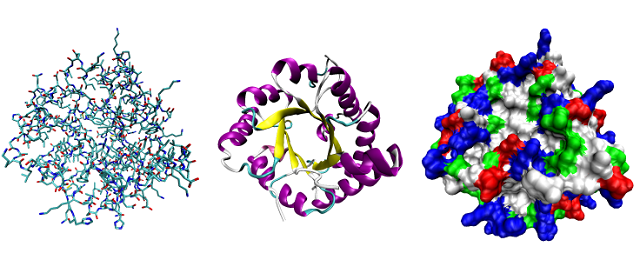
3. CHARACTER OF PROTEIN
1). Physical State:
All the proteins with the exception of crystalline few are amorphous colloids with no characteristic melting point [1, 2 &7].
2). Taste:
Proteins are generally tasteless. A few are bitter and they include proteoses, peptones, polypeptides and amino acids.
3). Effect of Heat:
Most proteins are irreversibly coagulated when heated. They undergo a change in solubility, molecular shape, size, biological activity and susceptibility to enzymes. That is why; we say that the proteins have been denatured.
4). Precipitation by Salts:
Different proteins are coagulated by the addition of different concentrations of suitable salts.
5). Viscosity:
Viscosity of proteins depends more upon the shape of the molecule and less on the size of the molecule. Less symmetrical is the molecule, greater will be its viscosity. Thus viscosity determines the blood pressure in the artries and other blood capillaries.
6). Optical Activity:
All the proteins possess optical activity due to presence of amino acids
as their constituents.
4. FUNCTION OF PROTEIN
Like carbohydrates and lipids, the proteins are important constituents of daily diet. They act as important body fuel. Their calorific value is 4 calories per g. When human food is deficient in carbohydrates which are instantaneous source of energy, the deficiency is made up protein mobilization.
1). Structural Proteins
These ar the structural constituents of the various cell, and tissues. Collagen, for example, is synthesized by fibroblasts and is the major extracellular protein of the connective tissue and bone. Glycoproteins form cell coats and walls [1& 6].
2). Storage Proteins
These store certain species after conjugation with them. For example ferritin stores iron in spleen, oval albumin, gliadin and zein are the storage proteins of egg, milk, wheat and corn respectively [2].
3). Contractile Proteins
Contractile proteins are the essential constituents of the contractile and motile systems. Mysoin is the contractile protein of the stationary filments in myofibrils and actin that of its moving filaments. Dynenin is the contractile protein of cilia and flagella [1].
4). Enzymes
Enzymes catalyse the biological reactions. A few examples are pepsin of stomach, alpha amylase of saliva, ribonuclease, cytochrome C, etc. Pepsin hydrolyses proteins in the stomach, alpha amylase degrades, starch in the mouth, ribonuclease hydrolyses RNA and cytochrome C transferts electrons to oxygen in respiration [2].
5). Protective Proteins
The protective proteins play an important role in the blood of vertebrates. Antibodies present in the blood defend the body against antigens, the proteins which after their entry inside the body cause a disease to process [9].
6). Transport Proteins
These proteins reversibly bind certain molecules and thus are capable of transporting them from one site to another via blood stream. For example, serum albumin binds free fatty-acids and transports them from liver to the adipose tissue [1&2].
7). Toxins
These are the proteins with toxic effects. Generally they are secreted by micro-organisms [7].
5. STRUCTURAL ORGANIZATION OF PROTEINS
Protein structure is normally described at four levels of organization.
1). Primary Structure
Primary structure is the linear sequence of amino acids held together by peptide bonds in its peptide chain. The peptide bonds form the backbone and side chains of amino acid residues project outside the peptide backbone. The free- 2H N group of the terminal amino acid is called as N-terminal end and the free · COOH end is called as C-terminal end. It is a tradition to number the amino acids from N-terminal end as No.1 towards the C-terminal end. Presence of specific Aminoacids at a specific number is very significant for a particular function of a protein. Any change in the sequence is abnormal and may affect the function and properties of protein [1, 2, 3 & 4].
2). Secondary Structure
The peptide chain thus formed assumes a three dimensional secondary structure by way of folding or coiling consisting of a helically-coiled, zig-zag, linear or mixed form. It results from the steric relationship between amino acids located relatively near each other in the peptide chain. The linkages or bonds involved in the secondary structure formation are hydrogen bonds and disulfide bonds [1, 5 &7].
a. Hydrogen bond
These are weak, low energy non-covalent bonds sharing single hydrogen by two electronegative atoms such as O and N.I
b. Disulphide bonds
These are formed between two cysteine residues. They are strong, high energy covalent bonds.
3). Tertiary Structure
The polypeptide chain with secondary structure mentioned above may be furtpher folded, super folded twisted about itself forming many sizes. Such a structural conformation is called tertiary structure. It is only one such conformation which is biologically active and protein in this conformation is called as native protein. Thus the tertiary structure is constituted by steric relationship between the amino acids located far apart but brought closer by folding. The bonds responsible for interaction between groups of amino acids are as follows [1, 6, 8 & 9]:
a. Hydrophobic interactions
Normally occur between nonpolar side chains of amio acids such as alanine, leucine, methionine, isoleucine and phenylalanine. They constitute the major stabilizing forces for tertiary structure forming a compact three-dimensional structure.
b. Hydrogen bonds
Normally formed by the polar side chains of the aminoacids.
c. Ionic or electrostatic interactions
These are formed between oppositely charged polar side chains of amino acids, such as basic and acidic amino acids.
d. Van der wall Forces
Occur between nonpolar side chains.
e. Disulfide bonds
These are the S-S bonds between SH groups of distant cysteine residues.
4). Quaternary Structure
Many proteins are made up of only one peptide chain. However, when a protein consists of two or more peptide chains held together by non-covalent interactions or by covalent cross-links, it is referred to as the quaternary structure. The assembly is often called as oligomer and each constituent peptide chain is called as monomer or subunit. The monomers of oligomeric protein can be identical or quite different in primary, secondary or tertiary structure.
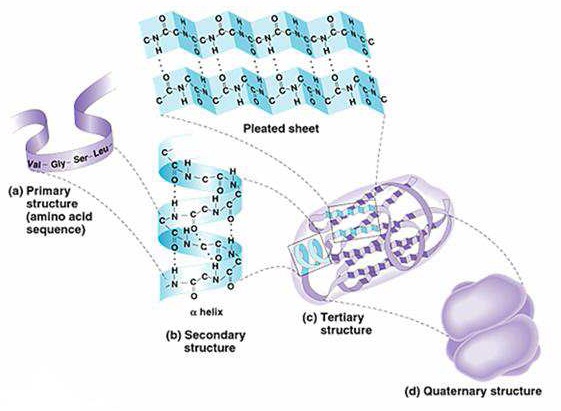
Examples: Protein with 2 monomers (dimer) is an enzyme called creatine phosphokinase (CPK) [1&2].
6. REFERENCES
2). M N Chaterjea, Medical Biochemistry, 7th Ed, Jaypee Brothers Medical Publishers, New Delhi, 2007.
4). Albert L Lehninger Principles of Biochemistry, 4th Ed, CBS Publisher, Delhi, 2004.
5). Lubert Stryer, Biochemistry, 5th Ed, W H Freeman and Company, 2002.
6). Pamela C Champe, Richard A Harvey, Illustrated Biochemistry, 4th Ed, J Lippincot Company, 2007.
7). HarperÂs Biochemistry, 26th Ed, Print-Hall, New Jersey, 2003.
8). M Rafiq, Biochemistry, The Carvan Book House, Lahore, 1st Ed.
9). Montogomary, Clinical Chemistry, the C V Mosby Company, 5th Ed.



