Natural killer (NK) cells, as a critical component of the innate immune system, exhibit a complex and dual role in the onset and progression of various cardiac diseases[1]. Among these conditions, autoimmune myocarditis (AM), a typical immune mediated myocarditis, represents an inflammatory myocardial disorder hallmarked by aberrant immune-mediated attacks on cardiac tissue, resulting in myocardial inflammation, cardiomyocyte injury, and potential progression to heart failure[2]. Dysregulated or aberrant activation of NK cells constitutes a key driver of myocardial inflammation and fibrosis[3-5].
This article reviews the role of NK cells in the pathogenesis of autoimmune myocarditis, the dual and context-dependent impacts of NK cells as both protective and pathogenic mediators in autoimmune myocarditis, the underlying mechanisms linking NK cell dysfunction to cardiac inflammation and fibrosis, how cytokines such as interferon gamma(IFN-γ) and Tumor Necrosis Factor Alpha(TNF-α) modulate NK cell activity in autoimmune myocarditis, and emerging evidence on NK cells in autoimmune myocarditis covering recent advances and unresolved questions.
Table of Contents
1. NK cells contribute to the pathogenesis of autoimmune myocarditis
2. NK cells are either protective or harmful in autoimmune myocarditis
3. NK cell dysfunction in autoimmune myocarditis: links to cardiac inflammation and fibrosis
4. Regulation of NK cell activity by cytokines including IFN-γ and TNF-α
5. Emerging evidence on NK cells in autoimmune myocarditis: recent advances and unresolved questions
01 NK cells contribute to the pathogenesis of autoimmune myocarditis
NK cells are large granular lymphocytes that mediate immune defense primarily through cytolytic activity and cytokine secretion, and are widely recognized as critical effectors in antiviral and antitumor immunity[6, 7]. However, their functional roles in myocarditis pathogenesis, particularly AM, are far more complex and context-dependent. In fact, NK cells initiate cytotoxicity via recognition of stress-induced activating ligands (e.g., MHC class I chain related proteins A/B, MICA/B) on target cells in conjunction with the absence of self-MHC class I molecules, a paradigm known as the "missing self" hypothesis, and in the setting of AM, cardiomyocytes upregulate these stress ligands in response to inflammatory signals or cellular damage, rendering them susceptible to NK cell-mediated attack. Emerging evidence suggests a dualistic role for NK cells in AM pathogenesis: while under certain autoimmune conditions NK cells exert protective immunoregulatory effects by directly eliminating activated autoreactive T cells or antigen-presenting cells (e.g., dendritic cells), thereby constraining the excessive activation of pathogenic CD4+/CD8+T cell populations[1], this regulatory function is critical for maintaining immune tolerance and preventing uncontrolled autoimmune responses[8]. Conversely, within specific myocardial microenvironments characterized by high concentrations of proinflammatory cytokines (e.g., interleukin 12(IL-12), interleukin 18(IL-18)) and low levels of immunosuppressive factors (e.g., transforming growth factor-β(TGF-β)), NK cells can adopt a pathogenic phenotype whereby they secrete copious amounts of IFN-γ, TNF-α, and granzyme B, which drive inflammatory cell infiltration into myocardial tissue and promote cardiomyocyte apoptosis, exacerbating disease progression.
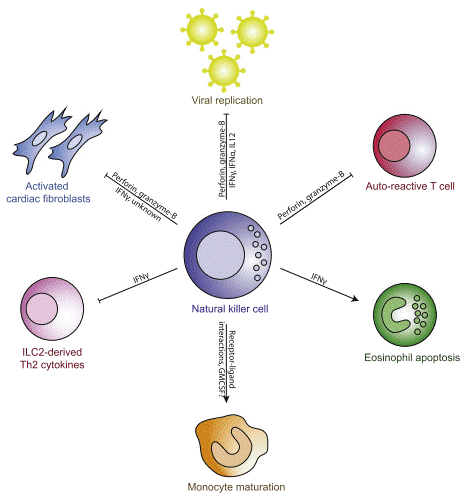
Fig. 1 Schematic of the mechanisms by which NK cells regulate the inflammatory cardiac environment[3].
02 NK cells are either protective or harmful in autoimmune myocarditis
NK cells function as a double-edged sword in the pathogenesis of autoimmune myocarditis[3, 9]. On one hand, NK cells contribute to the maintenance of systemic homeostasis by eliminating infected and neoplastic cells, with their cytotoxic activity and cytokine secretion representing critical pillars of innate immune defense[10]. Within the context of autoimmune disorders, NK cells may exert immunoregulatory effects by modulating the activity of adaptive immune cells, including T and B lymphocytes, thereby constraining aberrant autoimmune responses[11]. For instance, through engagement of the NKG2D/RAE-1 axis, NK cells can specifically recognize and eliminate overactivated autoreactive T cells, mitigating autoimmune-mediated tissue damage. Conversely, under specific pathological conditions, NK cells may directly or indirectly exacerbate myocardial inflammation and injury via the release of proinflammatory cytokines and cytotoxic mediators. In microenvironments characterized by high concentrations of IL-12 and IL-18, NK cells undergo robust activation and secrete copious amounts of IFN-γ, driving a Th1-skewed immune response that culminates in myocardial tissue damage[12, 13]. Emerging evidence indicates that NK cells play a central role in mediating proinflammatory responses during acute myocarditis following mRNA-based COVID-19 vaccination[9]. Furthermore, the direct cytotoxicity of NK cells toward cardiomyocytes, particularly when stress-induced upregulation of NKG2D ligands renders these cells susceptible to recognition, may induce cardiomyocyte death, thereby perpetuating a cycle of inflammation and fibrosis.
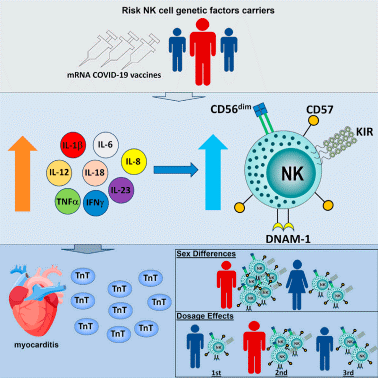
Fig. 2 The rapid onset of acute myocarditis following mRNA COVID-19 vaccinations remains unclear. Tsang et al. examined the pathological immune response, discovering that elevated NK cell activity and related genetic mechanisms significantly contribute to adverse vaccine events and heightened risk of vaccinerelated myocarditis[9].
03 NK cell dysfunction in autoimmune myocarditis: links to cardiac inflammation and fibrosis
The functional state of NK cells is tightly regulated by the balance between signals from their activating and inhibitory receptors. When this balance is perturbed, NK cell activation becomes dysregulated, contributing to the pathological progression of autoimmune diseases. NK cell dysfunction, including abnormal receptor profiles, dysregulated metabolic reprogramming, disrupted IFN-γ secretion, or impaired cytotoxic activity, diminishes their capacity to suppress autoreactive T cells, resulting in uncontrolled Th1/Th17 immune responses that further exacerbate myocardial injury. Beyond these direct pathogenic effects on myocardial tissue, NK cells also actively shape the inflammatory microenvironment that defines myocarditis. Myocardial inflammation is a defining hallmark of myocarditis, characterized by the infiltration of various immune cells, including macrophages, neutrophils, T cells, and B cells. In viral myocarditis, dysregulation of innate and adaptive immune responses is a key driver of myocardial injury and disease progression[14]. During the early stages of infection, NK cells activate macrophages via IFN-γ secretion. While this activation facilitates pathogen clearance, it may also induce "collateral damage" to macrophages during the clearance process, thereby further amplifying myocardial inflammation. Moreover, sustained and unresolved myocardial inflammation acts as a critical initiating stimulus for the development of myocardial fibrosis, a major pathological consequence of progressive myocarditis. Myocardial fibrosis is a common pathophysiological process in the progression of various cardiovascular diseases, characterized by the replacement of cardiomyocytes with collagen-rich scar tissue, which impairs cardiac function[15]. Activated fibroblasts (myofibroblasts), driven by molecular signals such as TGF-β, are key mediators of the fibrotic process. In AM, persistent inflammation and cellular injury can activate cardiac myofibroblasts, thereby promoting myocardial fibrosis. NK cells contribute to myocardial fibrosis through cytokine secretion, which directly or indirectly modulates fibroblast activation and collagen synthesis. For instance, granzyme B released by NK cells can promote fibroblast activation and accelerate the progression of myocardial fibrosis.
04 Regulation of NK cell activity by cytokines including IFN-γ and TNF-α
The immune cytokines network acts as a central regulator in controlling the phenotype and function of NK cells. The regulation of NK cell activity in autoimmune myocarditis by cytokines including IFN gamma ELISA, IL-6 and TNF alpha ELISA constitutes a highly elaborate immunoregulatory network, in which each cytokine exerts distinct yet interconnected effects on NK cell function, metabolic programming, and inflammatory outcomes. While IFN-γ and TNF-α primarily promote pro-inflammatory NK cell activation through synergistic signaling pathways, IL-6 adds further complexity by mediating both inflammatory amplification and potential regulatory feedback mechanisms. The temporal dynamics of cytokine expression establish a continuously evolving regulatory microenvironment throughout disease progression. The early stages of autoimmune myocarditis are characterized by predominant Th1 responses accompanied by high levels of IFN-γ and TNF-α production, which trigger robust NK cell activation and tissue infiltration[16]. However, with the persistence of inflammation, the upregulation of exhaustion markers such as TIM-3 on NK cells and potential skewing toward Th17/IL-6-dominated immune responses may impair NK cell functionality[17]. This temporal progression indicates that therapeutic strategies targeting specific cytokine signaling pathways require precise timing: early blockade of IFN-γ/TNF-α may alleviate initial tissue injury, whereas subsequent modulation of IL-6 signaling could target the chronic inflammatory components of the disease.
05 Emerging evidence on NK cells in autoimmune myocarditis: recent advances and unresolved questions
In recent years, substantial advances have been made in research investigating the roles of NK cells in AM.NK cells display marked subset heterogeneity. For instance, CD56bright NK cells are predominantly cytokine-secreting cells, whereas CD56dim NK cells are primarily defined by their cytotoxic effector functions. In AM models, an increased proportion of CD56bright NK cells correlates positively with disease activity, suggesting a potential proinflammatory role for this subset. Single-cell transcriptomic studies have revealed that NK cells in the peripheral blood and myocardial infiltrates of AM patients exhibit unique exhaustion-like characteristics, represented by a TOX+/TIGIT+/CD16– phenotype. This profile is accompanied by reduced IFN-γ secretion but sustained high expression of TNF-α, indicating that NK cells in AM may exist in a dysfunctional state. NK cells recognize stressed cardiomyocytes via the NKG2D–RAE-1 axis and mediate non-MHC-restricted cellular injury[18]. This signaling axis has been established as a critical pathogenic mediator in the mouse model of experimental autoimmune myocarditis (EAM).Studies have demonstrated that targeting NK cell metabolism, such as by inhibiting mTORC1 or enhancing mitochondrial oxidative phosphorylation, can restore the immunoregulatory functions of NK cells and reduce the severity of EAM[19], providing a novel therapeutic direction for AM. Preclinical studies have shown that low-dose IL-2 combined with TGF-β can selectively expand immunoregulatory NK cells (CD56brightCD9+), which significantly suppress the progression of EAM. Genetic modification of NK cells with chimeric antigen receptors (CAR-NK) represents a promising immunotherapeutic strategy[15, 20]. At present, CAR-NK approaches targeting cardiac-specific autoantigen peptide-MHC complexes are in the proof-of-concept stage and hold potential for precise immune intervention in AM. For example, CAR-NK cells targeting fibroblast activation protein (FAP) have been explored to alleviate cardiac fibrosis[15, 21], further highlighting the therapeutic potential of NK cells therapy.
In summary, the role of NK cells in autoimmune myocarditis is complex and dual-faced. While they can exert protective effects via immunoregulatory mechanisms, they may also aggravate disease progression under specific conditions through proinflammatory and cytotoxic pathways. A comprehensive understanding of the molecular mechanisms underlying NK cell dysfunction and their crosstalk with the cytokine network is critical for the development of effective immunotherapies against AM. Future studies will focus on the precise regulation of NK cell function, aiming to suppress autoimmune tissue damage while preserving their favorable immune surveillance properties.
Elabscience® Quick Overview of Popular Products
Table 1. Research Tools for autoimmune myocarditis
|
Cat. No. |
Product Name |
|
E-HSEL-H0002 |
High Sensitivity Human IL-2 (Interleukin 2) ELISA Kit |
|
E-HSEL-H0003 |
High Sensitivity Human IL-6 (Interleukin 6) ELISA Kit |
|
E-OSEL-H0001 |
QuicKey Pro Human IL-6(Interleukin 6) ELISA Kit |
|
CQH001 |
CellaQuant™ Human IL-6 (Interleukin 6) ELISA Kit |
|
ESP-H0009S |
Human IL-6 (Interleukin 6) solid ELISPOT Kit |
|
E-EL-H0150 |
Human IL-12(Interleukin 12) ELISA Kit |
|
E-EL-H0253 |
Human IL-18(Interleukin 18) ELISA Kit |
|
E-EL-H0109 |
Human TNF-α(Tumor Necrosis Factor Alpha) ELISA Kit |
|
E-HSEL-H0007 |
High Sensitivity Human IFN-γ (Interferon Gamma) ELISA Kit |
|
E-EL-H1617 |
Human GzmB(Granzyme B) ELISA Kit |
|
E-EL-H1587 |
Human TGF-β2(Transforming Growth Factor Beta 2) ELISA Kit |
|
XJH001 |
Human Th1/Th2 Flow Cytometry Staining Kit |
References:
[1] Liu, M., S. Liang, and C. Zhang, NK Cells in Autoimmune Diseases: Protective or Pathogenic? Frontiers in Immunology, 2021.12:624687.
[2] Leuschner, F., H.A. Katus, and Z. Kaya, Autoimmune myocarditis: Past, present and future. Journal of Autoimmunity, 2009. 33(3): p. 282-289.
[3] Ong, S., N.R. Rose, and D. Čiháková, Natural killer cells in inflammatory heart disease. Clinical immunology, 2017. 175: p. 26-33.
[4] Kumrić, M., et al., The Role of Natural Killer (NK) Cells in Acute Coronary Syndrome: A Comprehensive Review. Biomolecules, 2020. 10(11): p. 1514.
[5] He, X., et al., Inhibition of NK1.1 signaling attenuates pressure overload-induced heart failure, and consequent pulmonary inflammation and remodeling. Frontiers in Immunology, 2023.14:1215855.
[6] Björkström, N.K., B. Strunz, and H.-G. Ljunggren, Natural killer cells in antiviral immunity. Nature Reviews Immunology, 2022. 22(2): p. 112-123.
[7] Mace, E.M., Human natural killer cells: Form, function, and development. Journal of Allergy and Clinical Immunology, 2023. 151(2): p. 371-385.
[8] Roe, K., Immunoregulatory natural killer cells. Clinica Chimica Acta, 2024. 558: p. 117896.
[9] Tsang, H.W., et al., The central role of natural killer cells in mediating acute myocarditis after mRNA COVID-19 vaccination. Med, 2024. 5(4): p. 335-347.e3.
[10] Vivier, E., et al., Functions of natural killer cells. Nature Immunology, 2008. 9(5): p. 503-510.
[11] Fogel, L.A., W.M. Yokoyama, and A.R. French, Natural killer cells in human autoimmune disorders. Arthritis Research & Therapy, 2013. 15(4): p. 216.
[12] Grabie, N., et al., IL-12 is required for differentiation of pathogenic CD8+ T cell effectors that cause myocarditis. Journal of Clinical Investigation, 2003. 111(5): p. 671-680.
[13] Won, T., et al., Increased Interleukin 18-Dependent Immune Responses Are Associated With Myopericarditis After COVID-19 mRNA Vaccination. Frontiers in Immunology, 2022.13:851620.
[14] Golino, M., et al., Innate and adaptive immunity in acute myocarditis. International Journal of Cardiology, 2024. 404: p. 131901.
[15] Zheng, Q., et al., Fibroblast activation protein-targeted chimeric antigen-receptor-modified NK cells alleviate cardiac fibrosis. International Immunopharmacology, 2025. 157: p. 114760.
[16] Nagai, K., et al., Neutralizing the Th1 effector cytokines, IFN-γ and TNF-α, attenuates established experimental autoimmune anti-myeloperoxidase glomerulonephritis. Frontiers in Immunology, 2025.16:1589130.
[17] McCulloch, T.R., et al., Dichotomous outcomes of TNFR1 and TNFR2 signaling in NK cell-mediated immune responses during inflammation. Nature Communications, 2024. 15(1): p. 9871.
[18] Tong, Z., et al., Single-Cell Multi-Omics Identifies Specialized Cytotoxic and Migratory CD8+ Effector T Cells in Acute Myocarditis. Circulation, 2025. 152(14): p. 1003-1022.
[19] Verhezen, T., et al., Powering immunity: mitochondrial dynamics in natural killer cells. Trends in Molecular Medicine, 2026. 32(1): p. 49-64.
[20] Shin, E., et al., Understanding NK cell biology for harnessing NK cell therapies: targeting cancer and beyond. Frontiers in Immunology, 2023. 14:1192907.
[21] Zhang, Q., et al., Targeting cardiac fibrosis with Chimeric Antigen Receptor-Engineered Cells. Molecular and Cellular Biochemistry, 2024. 480(4): p. 2103-2116.



