Natural Killer (NK) cells exert a critical and multifaceted function in the pathogenesis of vascular inflammation and cardiac remodeling associated with hypertensive heart disease. These cells contribute to disease progression via direct cytotoxicity, modulation of inflammatory signaling cascades, complex crosstalk with other immune cell populations, and the secretion of potent pro inflammatory cytokines. Notably, NK cell activation is a key initiating event in this process, while NK cell depletion has emerged as a potential therapeutic strategy. Moreover, hypertensive heart disease with heart failure represents a severe clinical manifestation of the disease, where vascular inflammation and NK cell dysregulation play exacerbated roles.
This article reviews the primary contributions of NK cells to vascular inflammation in hypertensive heart disease, the mechanisms by which NK cells drive hypertensive heart disease through inflammatory signaling pathways, how interactions between NK cells, macrophages, and dendritic cells promote vascular inflammation, the roles of NK cell derived cytokines in mediating vascular inflammation in hypertension, and emerging evidence demonstrating that NK cell depletion alleviates vascular inflammation and cardiac remodeling in hypertensive mice.
Table of Contents
1. Primary contribution of NK cells to vascular inflammation in hypertensive heart disease
2. NK cells drive hypertensive heart disease via inflammatory signaling pathways
3. NK cell interactions with macrophages and dendritic cells promote vascular inflammation
4. NK cell derived cytokines driving vascular inflammation in hypertension
5. NK cell depletion reduces vascular inflammation and cardiac remodeling in hypertensive mice
01 Primary contribution of NK cells to vascular inflammation in hypertensive heart disease
NK cells are essential components of the innate immune system, which can rapidly respond to cellular perturbations and modulate both innate and adaptive immune responses[1, 2]. As the classic phenotypic markers of these cells, CD16 CD56 natural killer cells are the major subset involved in cardiovascular inflammation. Under hypertensive conditions, these cells infiltrate perivascular adipose tissue and arterial walls, thereby directly contributing to vascular inflammation. Such infiltration results in endothelial dysfunction, proliferation of vascular smooth muscle cells, and recruitment of other leukocytes, all of which represent hallmark pathological features of vascular injury in hypertension. Accumulating evidence indicates that inflammation serves as a pivotal mediator in the vascular remodeling and cardiac dysfunction associated with hypertension. Chronic hypertension is characterized by low grade systemic inflammation within the cardiovascular system, where infiltration of immune cells, particularly macrophages, triggers inflammatory cascades that promote vascular and ventricular remodeling. Furthermore, NK cells participate in maintaining cardiac homeostasis by suppressing the maturation and trafficking of inflammatory cells, whereas their dysregulation under pathological injury can aggravate inflammatory responses[3].
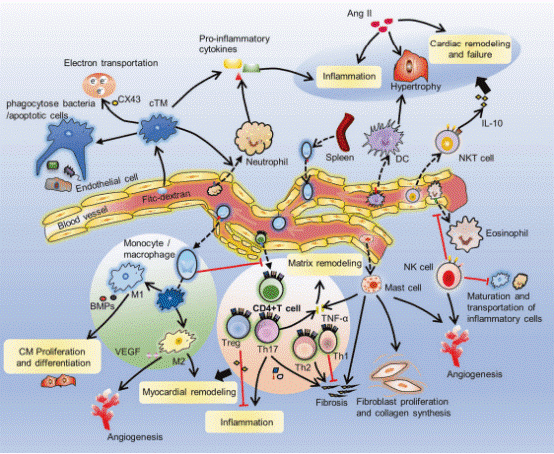
Fig. 1 Immune cells in the heart. Resident NK cells reduce cardiac eosinophil infiltration. NK cells prevent the maturation and transport of inflammatory cells[3].
02 NK cells drive hypertensive heart disease via inflammatory signaling pathways
NK cells contribute to hypertensive heart disease through the activation of key inflammatory signaling pathways, most notably NLRP3, STAT1, and the NF-κB inflammasome. The NLRP3 inflammasome is a multiprotein complex central to innate immune responses that, upon activation, mediates caspase 1 activation and the release of pro inflammatory cytokines including interleukin-1β (IL-1β) and interleukin-18 (IL-18)[4-8]. which can be quantitatively assessed using caspase-1 activation assay and NLRP3 inflammasome assay. Mechanical stress imposed on the heart, such as that induced by elevated blood pressure, triggers inflammatory activation and the development of hypertrophic heart disease, in which IL-1β exerts a critical role in promoting cardiac hypertrophy and heart failure[9]. NK cells can be activated by interleukin-12 (IL-12) and IL-18 secreted by antigen presenting cells, resulting in sustained interferon-γ (IFN-γ) production which in turn activates STAT1 to promote the polarization of macrophages toward a pro inflammatory M1 phenotype and upregulate the expression of MHC class I molecules on cardiomyocytes. These processes are typically evaluated by IFN-γ assay. This establishes a vicious cycle in which NK cell activation propagates inflammatory cascades that further aggravate cardiac pathology. Activation of the NLRP3 inflammasome represents a key mechanism underlying hypertension induced target organ damage[10]. In the setting of hypertension, aberrant activation of NF-κB plays a pivotal role in mediating cardiovascular remodeling and injury. NF-κB drives the transcriptional expression of pro inflammatory cytokines, including tumor necrosis factor α (TNF-α), interleukin-6 (IL-6), and IL-1β, as well as adhesion molecules such as vascular cell adhesion molecule-1 (VCAM-1) and intercellular adhesion molecule-1 (ICAM-1), and chemokines, with TNF-α levels commonly measured by TNF-α assay. These mediators promote the recruitment of natural killer (NK) cells and sustain their activation within the pressure overloaded myocardium. Collectively, the involvement of these signaling pathways highlights the mechanisms by which NK cells shape the pro inflammatory microenvironment characteristic of hypertension.
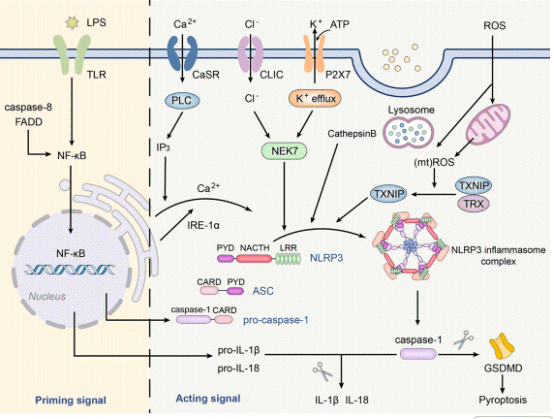
Fig. 2 Canonical activation of the NLRP3 inflammasome. The NLRP3 inflammasome comprises NLRP3 protein, ASC, and pro-caspase-1[5].
03 NK cell interactions with macrophages and dendritic cells promote vascular inflammation
The interplay between NK cells and other immune cells, particularly macrophages and dendritic cells (DCs), is crucial for promoting vascular inflammation in hypertension. Macrophages are abundant immune cells in tissues that exhibit plasticity and can polarize into different phenotypes, with M1 macrophages generally being pro inflammatory[12]. NK cells engage in bidirectional IFN-γ, TNF-α crosstalk with macrophages. This interaction can lead to enhanced pro inflammatory responses. Macrophage infiltration initiates inflammatory processes contributing to vascular remodeling in hypertension. Dectin1, for example, contributes to hypertensive vascular injury by promoting macrophage infiltration through the activation of the Syk/NF-κB pathway. NK cells also interact with dendritic cells through mechanisms such as NKG2D Rae 1 and CD40, CD40L interactions. These interactions enhance DC maturation and IL-12 secretion, which in turn amplifies T cell priming and vascular inflammation. Additionally, NK cell function is regulated by surface receptors such as nkg2a (an inhibitory receptor) and nkp46 (an activating receptor), which fine tune nk cell activation and subsequent inflammatory responses. Dendritic cells, alongside monocytes/macrophages, contribute to blood pressure elevation via effects on kidney and vascular function[13]. Plasmacytoid dendritic cells, another subset of DCs, also contribute to arterial endothelial dysfunction and inflammation in renovascular hypertension[14]. The coordinated actions of NK cells, macrophages, and dendritic cells thus create a complex immune network that sustains and amplifies the inflammatory processes central to hypertensive heart disease.
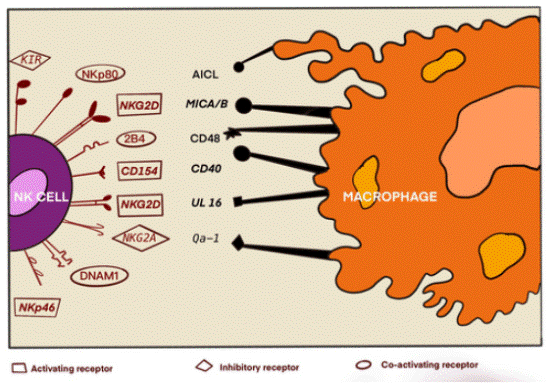
Fig. 3 Ligands and receptors involved in the interplay of macrophages and natural killer cells. Different receptors, encompassing both activating and inhibitory NK cell receptors, engage with macrophages via ligands located on their cell surfaces[15].
04 NK cell derived cytokines driving vascular inflammation in hypertension
NK cells are potent producers of various cytokines and chemokines[15]. Among these, NK cell derived IFN-γ has been identified as a dominant cytokine that drives vascular inflammation. IFN-γ upregulates VCAM-1 and ICAM-1 expression on endothelial cells, thereby facilitating monocyte adhesion. This process is critical for the recruitment of immune cells into the vascular wall, which contributes to vascular inflammation and injury. Additionally, IFN-γ induces inducible nitric oxide synthase (iNOS) in macrophages and stimulates collagen deposition in cardiac fibroblasts, further promoting vascular and cardiac remodeling. The capacity of NK cells to secrete IFN-γ and other inflammatory cytokines also plays an important role in the onset of acute myocardial infarction and the regulation of pathological cardiac remodeling[16]. In addition to IFN-γ, TNF-α and granulocyte macrophage colony stimulating factor (GM-CSF) further enhance leukocyte infiltration and vascular remodeling. TNF-α, a master pro inflammatory cytokine, orchestrates vascular responses by activating endothelial cells, rendering blood vessels "leaky and sticky" and inducing the expression of adhesion molecules, thereby facilitating leukocyte infiltration. The role of cytokines in regulating the structure and function of blood vessels in hypertension is well established, as they contribute to the establishment of a pro inflammatory milieu[17]. Elevated cytokine levels, along with oxidative stress, are characteristic features of hypertension and contribute to the associated cardiovascular damage[12].
05 NK cell depletion reduces vascular inflammation and cardiac remodeling in hypertensive mice
Experimental evidence strongly supports a causal role of NK cells in hypertensive vascular inflammation and cardiac remodeling. In animal models of hypertension induced by angiotensin II (Ang II) or deoxycorticosterone acetate (DOCA) salt, NK cell depletion mediated by anti NK1.1 significantly attenuates adverse cardiovascular outcomes. Specifically, NK cell depletion results in reduced aortic inflammation, decreased perivascular fibrosis, lowered systolic blood pressure, and ameliorated left ventricular hypertrophy and diastolic dysfunction. This demonstrates that NK cells are not merely correlated with hypertension but actively contribute to its pathological progression. Additionally, inhibition of NK1.1 signaling has been shown to attenuate pressure overload induced heart failure, as well as the consequent pulmonary inflammation and remodeling[18]. Collectively, these findings underscore the therapeutic potential of targeting NK cells or their associated signaling pathways to mitigate the detrimental effects of hypertension on the cardiovascular system.
In summary, this systematic review delineates the pivotal roles of NK cells in vascular inflammation and pathological cardiac remodeling in hypertensive heart disease, a cardiovascular disorder driven by chronic low grade inflammation. It outlines the multifaceted mechanisms by which NK cells promote hypertensive vascular injury, including infiltration into perivascular and arterial tissues, activation of inflammatory signaling pathways such as NF-κB and NLRP3 inflammasome, and crosstalk with macrophages and dendritic cells that amplifies pro inflammatory responses. The review also highlights the key functions of NK cell derived cytokines, especially IFN-γ, TNF-α, and GM-CSF, in mediating endothelial dysfunction, leukocyte recruitment, and vascular remodeling. Furthermore, preclinical studies show that NK cell depletion attenuates aortic inflammation, perivascular fibrosis, and cardiac hypertrophy in hypertensive mouse models, supporting a causal role for NK cells in disease progression. Collectively, these insights advance the understanding of immune mediated hypertensive cardiovascular damage and identify NK cells as potential therapeutic targets for hypertensive heart disease.
Elabscience® Quick Overview of Popular Products:
Table 1. Research Tools for hypertensive heart disease
|
Cat. No. |
Product Name |
|
XJM003 |
Mouse Bone Marrow-derived Dendritic Cells (BMDC) Induction and Identification Kit |
|
XJM004 |
RAW 264. 7 Polarized M1 Macrophage Induction and Identification Kit |
|
MIM005N |
EasySort™ Mouse NK Cell Isolation Kit |
|
E-AB-FC0007 |
Anti-Human CD3-FITC/CD19-APC/CD16+CD56-PE Cocktail |
|
E-HSEL-M0002 |
High Sensitivity Mouse IL-4 (Interleukin 4) ELISA Kit |
|
E-HSEL-H0003 |
High Sensitivity Human IL-6 (Interleukin 6) ELISA Kit |
|
E-HSEL-H0005 |
High Sensitivity Human IL-10 (Interleukin 10) ELISA Kit |
|
E-EL-H0081 |
Human GM-CSF(Granulocyte-Macrophage Colony Stimulating Factor) ELISA Kit |
|
E-EL-H0108 |
Human IFN-γ(Interferon Gamma) ELISA Kit |
|
E-EL-H0109 |
Human TNF-α(Tumor Necrosis Factor Alpha) ELISA Kit |
|
E-EL-H0149 |
Human IL-1β(Interleukin 1 Beta) ELISA Kit |
|
E-EL-H0150 |
Human IL-12(Interleukin 12) ELISA Kit |
|
E-EL-H0253 |
Human IL-18(Interleukin 18) ELISA Kit |
|
E-EL-H0326 |
Human Ang-Ⅱ(Angiotensin Ⅱ) ELISA Kit |
|
E-EL-H0753 |
Human NOS2/iNOS(Nitric Oxide Synthase 2, Inducible) ELISA Kit |
|
E-EL-H2557 |
Human NLRP3(NACHT, LRR and PYD domains-containing protein 3) ELISA Kit |
References:
[1] Jiang, H. and J. Jiang, Balancing act: the complex role of NK cells in immune regulation. Frontiers in Immunology, 2023. 14:1275028.
[2] Felkle, D. , et al. , The immunomodulatory effects of antihypertensive therapy: A review. Biomedicine & Pharmacotherapy, 2022. 153: p. 113287.
[3] Sun, K. , Y. -y. Li, and J. Jin, A double-edged sword of immuno-microenvironment in cardiac homeostasis and injury repair. Signal Transduction and Targeted Therapy, 2021. 6(1):79.
[4] Jafari, N. , et al. , Crosstalk of NLRP3 inflammasome and noncoding RNAs in cardiomyopathies. Cell Biochemistry and Function, 2023. 41(8): p. 1060-1075.
[5] Chen, Y. , et al. , The NLRP3 inflammasome: contributions to inflammation-related diseases. Cellular & Molecular Biology Letters, 2023. 28(1):51.
[6] Chen, M. -y. , et al. , The Signaling Pathways Regulating NLRP3 Inflammasome Activation. Inflammation, 2021. 44(4): p. 1229-1245.
[7] Toldo, S. , et al. , Targeting the NLRP3 inflammasome in cardiovascular diseases. Pharmacology & Therapeutics, 2022. 236: p. 108053.
[8] Yao, J. , et al. , The role of inflammasomes in human diseases and their potential as therapeutic targets. Signal Transduction and Targeted Therapy, 2024. 9(1):10.
[9] Higashikuni, Y. , et al. , NLRP3 inflammasome activation through heart-brain interaction initiates cardiac inflammation and hypertrophy during pressure overload. Circulation, 2023. 147(4): p. 338-355.
[10] Liao, X. , et al. , Targeting the NLRP3 inflammasome for the treatment of hypertensive target organ damage: Role of natural products and formulations. Phytotherapy Research, 2023. 37(12): p. 5622-5638.
[11] Hussain, M. S. , et al. , Probing the links: Long non-coding RNAs and NF-κB signalling in atherosclerosis. Pathology - Research and Practice, 2023. 249: p. 154773.
[12] Martínez-Casales, M. , R. Hernanz, and M. J. Alonso, Vascular and macrophage heme oxygenase-1 in hypertension: a mini-review. Frontiers in Physiology, 2021. 12:643435.
[13] Wenzel, U. O. , H. Ehmke, and M. Bode, Immune mechanisms in arterial hypertension. Recent advances. Cell and Tissue Research, 2021. 385(2): p. 393-404.
[14] Srinivas, B. , et al. , Role of plasmacytoid dendritic cells in vascular dysfunction in mice with renovascular hypertension. Heliyon, 2024. 10(11).
[15] Hooda, V. and A. Sharma, Interactions of NK cells and macrophages: from infections to cancer therapeutics. Immunology, 2024. 174(3): p. 287-295.
[16] Ortega-Rodríguez, A. C. , et al. , Altered NK cell receptor repertoire and function of natural killer cells in patients with acute myocardial infarction: a three-month follow-up study. Immunobiology, 2020. 225(3): p. 151909.
[17] dos Passos, R. R. , et al. , Immunomodulatory activity of cytokines in hypertension: a vascular perspective. Hypertension, 2024. 81(7): p. 1411-1423.
[18] He, X. , et al. , Inhibition of NK1.1 signaling attenuates pressure overload-induced heart failure, and consequent pulmonary inflammation and remodeling. Frontiers in Immunology, 2023. 14:1215855.



