RAW 264.7 is the go‑to cell line for inflammation and immunology research. It’s stable, easy to culture, and behaves like primary macrophages, making it the gold standard for anti‑inflammatory drug screening and mechanistic studies.
So how do you actually set up an LPS‑induced inflammation model and measure cytokines? Let’s get into it.
Table of Contents
1. Basic characteristics of RAW 264.7
2. Steps and key points for establishing a validation model using RAW 264.7
3. Cytokine detection: case examples
4. Recommended products for cytokine detection
01 Basic characteristics of RAW 264.7
RAW 264.7 (mouse monocyte‑macrophage leukemia cell line) was derived from an Abelson MuLV‑induced tumor in a Balb/c mouse. This immortalized line is easy to grow and expand. It also mimics complex in vivo immune environments, a key reason it’s widely used in macrophage biology, inflammation, and immune regulation.
Key features
Easy culture
Grows rapidly; no tricky isolation like primary macrophages.
Macrophage‑like
Adherent, phagocytic, and positive for F4/80 and CD11b.
Polarization capacity
Under specific stimuli, RAW 264.7 can differentiate into:
M1 (classical activation): Induced by LPS + IFN-γ, pro-inflammatory, antimicrobial, antitumor
M2 (alternative activation): Induced by IL-4 or IL-13, tissue repair, immunomodulation
This makes it a core in vitro model for studying macrophage polarization, inflammatory control, and related diseases (e.g., cancer, osteoporosis, atherosclerosis).
Immune response
Expresses Toll-like receptors (especially TLR4). Upon LPS stimulation, it secretes key inflammatory cytokines such as TNF-α, IL-6, and IL-1β[1].
02 Steps and key points for establishing a validation model using RAW 264.7
Inflammation is a protective response to injury or infection, but chronic or excessive inflammation is closely linked to autoimmune diseases, metabolic disorders, neurodegenerative diseases, and cancer[2-3].
In cell biology research, LPS is the most commonly used pro-inflammatory inducer due to its potent inflammatory properties.
Experimental protocol
Cell culture
Cell line: RAW 264.7 (confirm good condition, no contamination)
Medium: DMEM (high glucose) + 10% FBS + 1% penicillin/streptomycin
Thawing & passaging: Use cells after at least 3 passages to ensure stability
Plating (for a 12-well plate):
Density: 2-5 × 105 cells/mL, 500 μL per well, allow overnight adhesion (12-16 hours)
Inflammation induction
Stimulant: LPS, typically 0.1-1 μg/mL
Other options: PMA, IFN-γ, GM-CSF, TNF-α (often combined with LPS to enhance response)
Treatment time: 12-48 hours
Detection
ELISA: Collect cell supernatant to measure secreted TNF-α, IL-6, IL-1β, etc.
03 Cytokine detection: case examples
The LPS-induced RAW 264.7 inflammation model primarily activates the TLR4-NF-κB/MAPK signaling pathway[4], leading to robust expression of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β) as well as NO and iNOS.
Studies show that after stimulation with 1.0 μg/mL LPS, RAW 264.7 cells significantly upregulate IL-6, IL-1β, and TNF-α, while IL-10 (an anti-inflammatory cytokine) remains low. Treatment with BiOSe or BiOSe-RA reduces IL-6, IL-1β, and TNF-α levels and increases IL-10, with the BiOSe-RA group showing the most pronounced effect[1].
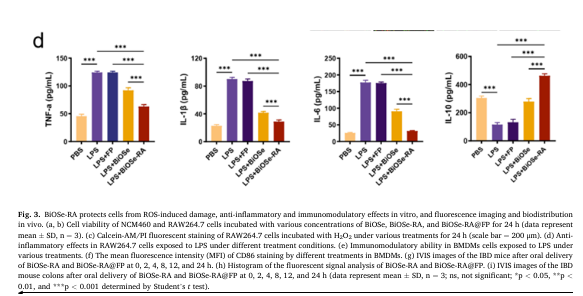
Fig. 1 Cytokine secretion in LPS-stimulated RAW 264.7 cells.
04 Recommended products for cytokine detection
Table 1. Related ELISA products for cytokine detection
|
Cat. No. |
Product Name |
|
CQM003 |
CellaQuant™ Mouse IL-1β (Interleukin 1 Beta) ELISA Kit |
|
CQM001 |
CellaQuant™ Mouse IL-6 (Interleukin 6) ELISA Kit |
|
CQM004 |
CellaQuant™ Mouse IL-10 (Interleukin 10) ELISA Kit |
|
CQM002 |
CellaQuant™ Mouse TNF-α (Tumor Necrosis Factor Alpha) ELISA Kit |
Facing something unusual with your RAW 264.7 cultures? A quick comment could save a colleague hours of troubleshooting.
References:
[1] Yao J, Yang Y, Zhao H, et al. Bi2O2Se nanosheets coated with rosmarinic acid for colon-targeted treatment of inflammatory bowel disease[J]. Chemical Engineering Journal, 2025, 506(000). DOI: 10.1016/j.cej.2025.160230.
[2] He T, Zhou B, Sun, et al. The bone-liver interaction modulates immune and hematopoietic function through Pinch-Cxcl12-Mbl2 pathway[J]. Cell Death and Differentiation, 2024, 31(1):90-105. DOI: 10.1038/s41418-023-01243-9.
[3] Yacine A, Ali M Z, Alharbi A B, et al. Chronic Inflammation: A Multidisciplinary Analysis of Shared Pathways in Autoimmune, Infectious, and Degenerative Diseases[J]. Cureus, 2025. DOI: 10.7759/cureus.82579.
[4] Qin Y, Li K, Zhang Q, et al.Linoleic acid inhibits lipopolysaccharide-induced inflammation by promoting TLR4 regulated autophagy in murine RAW 264.7 macrophages[J]. Journal of Applied Biomedicine, 2024, 22(4). DOI: 10.32725/jab.2024.023.



