In cell-based assays, the CCK-8 method is commonly used for analyzing cell proliferation and cytotoxicity. In this article, we have compiled a list of frequently asked questions about CCK-8 and analyzed abnormal results based on real-world cases, hoping to help you achieve satisfactory experimental outcomes.
Table of Contents
1. Principle of the CCK-8 cell viability assay
2. Comparative analysis of common cell viability assays
3. Representative experimental results using CCK-8
4. Frequently asked questions about CCK-8 assay protocols
5. Troubleshooting abnormal results in CCK-8 assays
01 Principle of the CCK-8 cell viability assay
The pink-colored WST-8 is reduced by intracellular dehydrogenases in the presence of the electron carrier 1-Me-PMS, producing a yellow formazan product. This product exhibits a characteristic absorption peak at a wavelength of 450 nm. The rate of change in absorbance (i.e., the rate of formazan production) is proportional to dehydrogenase activity, which indirectly reflects the number of viable cells. This characteristic can be used directly for analyzing cell proliferation and cytotoxicity.
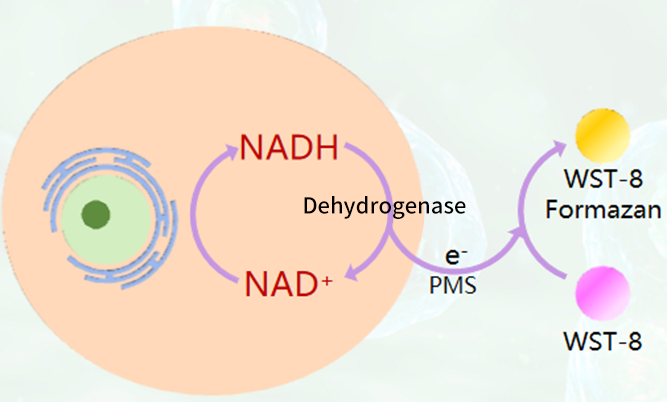
Fig. 1 CCK-8 detection principle.
02 Comparative analysis of common cell viability assays
Table 1. Performance comparison of four common cell proliferation/cytotoxicity assays: MTT, XTT, WST-1, and CCK-8
|
Method |
XTT |
WST-1 |
||
|
Solubility of formazan |
Insoluble |
Water-soluble |
Water-soluble |
Water-soluble |
|
Form |
Powder |
Liquid |
Liquid |
Liquid |
|
Usage |
Use after dissolution |
Prepare in advance |
Use directly |
Use directly |
|
Sensitivity |
+ |
++ |
++ |
++ |
|
Detection speed |
+ |
++ |
++ |
+++ |
|
Detection range |
560-600 nm |
420-480 nm |
420-480 nm |
430-490 nm |
|
Cytotoxicity |
+ |
- |
- |
-- |
|
Stability |
+ |
- |
+ |
++ |
|
Reproducibility |
+ |
++ |
+++ |
+++ |
|
Convenience |
+ |
++ |
++ |
+++ |
From the comparison of different detection methods for cell proliferation and cytotoxicity analysis, it can be seen that CCK-8 outperforms MTT in terms of water solubility, sensitivity, detection speed, and cytotoxicity.
03 Representative experimental results using CCK-8
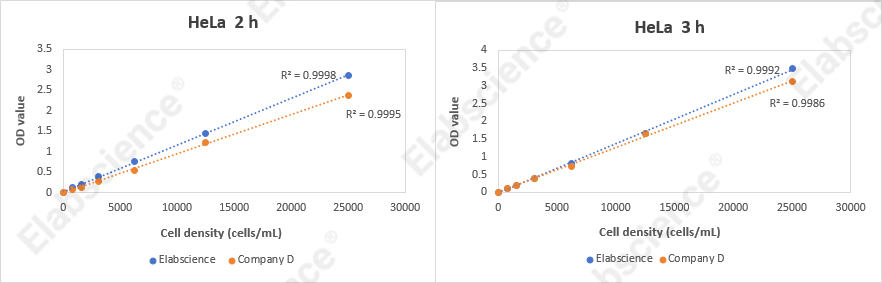
Fig. 2 CCK-8 assay experimental results. HeLa cells at different densities (25,000, 12,500, 6,250, 3,125, 1,562, 780, 390, and 0 cells/mL) were seeded into a 96-well plate and cultured at 37°C for 24 hours. Subsequently, they were incubated with the Enhanced Cell Viability Assay Kit (CCK-8) (E-CK-A362) for 2 hours and 3 hours, respectively, and the OD values were measured at a wavelength of 450 nm.
Example of Result Analysis
Table 2. Cell viability analysis of different experimental groups
|
Group |
OD Value |
Average |
Subtract blank |
Cell viability |
||
|
Blank control |
0.1986 |
0.2049 |
0.2052 |
0.2029 |
/ |
/ |
|
Negative control |
1.2725 |
1.2655 |
1.2058 |
1.2479 |
1.0450 |
100% |
|
Group 1 |
0.9847 |
0.9554 |
1.0351 |
0.9917 |
0.7888 |
75.48% |
|
Group 2 |
0.9079 |
0.9543 |
0.9238 |
0.9287 |
0.7258 |
69.45% |
|
Group 3 |
0.8154 |
0.862 |
0.8398 |
0.8391 |
0.6362 |
60.88% |
|
Group 4 |
0.6335 |
0.6345 |
0.6638 |
0.6439 |
0.4410 |
42.20% |
Cell viability (%) = (OD sample – OD blank) / (OD control– OD blank) × 100%
OD Sample: OD value of the experimental group well
OD Control: OD value of the negative control well
OD Blank: OD value of the blank control well
04 FAQs about CCK-8 assay protocols
Q1: How many cells should be added per well in a CCK-8 assay?
A: The number of cells per well depends on factors such as cell size and proliferation rate. Typically, for cell proliferation assays, 100 μL of cell suspension containing approximately 2,000 cells is added per well. For cytotoxicity assays, 100 μL containing approximately 5,000 cells is added per well.
Q2: How should the incubation time after CCK-8 addition be determined?
A: Add 10 μL of CCK-8 solution per well and continue incubating for 1-4 hours (the incubation conditions for CCK-8 are the same as those for cell culture).
The exact time depends on the cell type, cell number, and metabolic activity.
Guideline: Observe every half hour. When the color of the control wells (without drug) turns orange-yellow (visibly noticeable color change), the reaction can be terminated. Insufficient time may result in weak signals, while excessive time may cause OD values to saturate, masking drug inhibition effects.
Q3: How should CCK-8 reagent be stored? Does repeated freeze-thawing affect it?
A: CCK-8 should be stored protected from light at –20°C or 4°C. For long-term storage, –20°C is recommended. For short-term frequent use, the reagent can be aliquoted and stored at 4°C protected from light (use within 1–2 weeks). Repeated freeze-thawing reduces reagent sensitivity. It is recommended to aliquot the reagent into single-use portions after the first opening to avoid repeated freeze-thaw cycles.
Q4: Why are the results from the edge wells of a 96-well plate often abnormal?
A: This is due to the "edge effect." The outermost wells of a 96-well plate (rows A and H, columns 1 and 12) experience faster evaporation, leading to concentrated culture medium and differences in cell status.
Solution: Fill the outermost wells with sterile PBS or culture medium only, without seeding cells, to isolate the edge environment.
Q5: What is the ideal range for OD values in a CCK-8 assay?
A: Typically, OD values should fall between 0.2 and 1.5. If the OD value is too high (>2.0), the cells may be too dense or the color development time may be too long. If the OD value is too low (<0.2), the cell number may be too low or the drug toxicity may be too strong.
Guideline: The OD value of the control wells (without drug) is usually around 0.8-1.2 for optimal results.
Q6: If the drug itself has color or is easily oxidized, will it affect the results?
A: Yes, this is a common issue. It is recommended to set up a background control well containing "no cells + drug + CCK-8." During calculation, subtract the OD of the corresponding background well from the experimental well OD to eliminate interference from the drug's color or reducing substances.
Q7: Can CCK-8 be recycled and reused?
A: Reuse is not recommended. Although color development is an enzymatic reaction, the presence of metabolic activity from viable cells in the system can cause significant errors upon reuse and increase the risk of contamination.
Q8: How should a blank control be set up?
A: Wells containing the corresponding amount of cell culture medium and CCK-8 but without cells can be used as blank controls. If there is concern that the drug used may interfere with detection, set up blank control wells containing the corresponding amount of cell culture medium, drug, and CCK-8 but without cells.
Q9: Why do blank wells containing only CCK-8 (no cells) show an increase in OD value?
A: During the experiment, if cells are cultured for an extended period, evaporation may occur. The reaction environment is warm, which can cause some liquid to evaporate. Although there are no cells in the blank wells, as the liquid evaporates, the concentration of the drug (if present) effectively increases, causing slight changes in the blank control OD value (though not significantly). If you are concerned about blank control values, add culture medium, water, or PBS to the outer ring of the 96-well plate to maintain humidity.
Additionally, placing the 96-well plate near the water reservoir inside the incubator can help reduce evaporation. Filling the two outer rings of the 96-well plate with PBS can minimize the impact on internal wells and blank controls, as evaporation will occur first from the periphery.
Q10: How often should OD values be measured?
A: During preliminary experiments, measure OD values at 0.5, 1, 2, and 4 hours using a microplate reader, then select a time point within a suitable absorbance range for subsequent experiments.
Q11: If OD values cannot be measured immediately after adding CCK-8, how can the samples be temporarily preserved? How long can they be stored?
A: If OD measurement needs to be delayed, add 10 μL of 0.1 M HCl solution or 1% w/v SDS solution to each well, store protected from light at room temperature, and the absorbance will remain stable for up to 24 hours.
Q12: If the culture medium color or pH has changed, is it necessary to replace it with fresh medium?
A: When adding CCK-8, if the cells have been cultured for an extended period and the medium color or pH has changed, it is recommended to replace it with fresh culture medium.
Q13: What types of cells are suitable for CCK-8 assays?
A: Since the assay measures mitochondrial enzyme activity, any eukaryotic cells containing mitochondria are suitable, such as single cells and animal cells. However, cells with cell walls, such as plant cells and yeast cells, are not recommended due to low substrate entry efficiency.
Q14: Can CCK-8 replace the BrdU or EdU method for detecting cell proliferation?
A: CCK-8 reflects cell viability by detecting dehydrogenase activity in cells during the growth phase, whereas thymidine analog incorporation methods (BrdU/EdU) detect cell viability by incorporating nucleotide analogs into synthesized DNA. These methods are correlated to some extent. CCK-8 can replace thymidine incorporation methods for detecting cell proliferation and is more convenient to perform.
Q15: When two cell types are co-cultured (e.g., bone marrow mesenchymal stem cells and suspension T cells), and after a period of co-culture, only the growth and proliferation of the adherent mesenchymal stem cells need to be measured, can CCK-8 be used? How should it be done?
A: For co-culture systems, the approach depends on the specific situation:
If measuring the growth of adherent cells, the suspension T cells can be removed, and the standard CCK-8 protocol can be used.
If the T cells are critical for mesenchymal stem cell growth and cannot be removed, the measured viability will reflect the total activity. In this case, the number of T cells in each well needs to be estimated. If the total number of T cells is similar across conditions or if the T cell number is minimal compared to mesenchymal stem cells, the assay can still be performed, and the effect of T cells can be accounted for using control wells.
05 Troubleshooting abnormal results in CCK-8 assays
Scenario 1: After drug treatment, cell condition deteriorates, but the OD value of the drug-treated group is higher than that of the control group.
|
Reason |
Solution |
|
The drug has redox activity |
Set up background control wells containing "no cells + drug + CCK-8" |
Scenario 2: Under the microscope, the cells appear mostly dead, but the measured OD value is still high.
|
Reason |
Solution |
|
Plate contamination |
Visually observe whether the plate is contaminated |
|
Cell seeding density is too high |
Reduce the cell seeding density |
|
CCK-8 incubation time is too long |
Reduce the CCK-8 incubation time |
Scenario 3: The OD values of all wells are around 0.3, with no color development.
|
Situation |
Reason |
Solution |
|
Under the microscope, the control group cells appear to be in good condition, but the cell density in both the control wells and drug-treated wells is relatively low |
Low seeding density, and the proliferation rate has not reached the detection sensitivity |
Increase the seeding density |
|
The control group cells are in good condition, and a trend is observed under the microscope between the drug-treated group and the untreated group |
The culture medium has redox activity |
Replace the culture medium and re-test |
|
Under the microscope, the control group cells are in poor condition with extensive death |
The seeding density is too high, causing the cells to become overconfluent, leading to crowding-induced death and contact inhibition |
Reduce the cell seeding density |
|
The cell condition in both the control group and the drug-treated group is poor, and all groups are essentially the same. |
The cell condition was already poor before seeding |
Improve the cell condition before seeding |



