Atherosclerosis is the most common pathological process underlying cardiovascular disease (CVD), encompassing myocardial infarction and stroke, the leading causes of mortality in the western hemisphere. For a long time, atherosclerosis was considered an inevitable age related disorder marked by the passive accumulation of lipids within the vascular wall[1]. The pathogenesis of CVD is multifactorial, involving genetic predisposition and various modifiable risk factors such as smoking, hypercholesterolemia, diabetes, and hypertension. Notably, accumulating evidence has established a critical role of the immune system in atherogenesis, with both inflammatory mediators and autoimmune components including autoantibodies, autoantigens, and autoreactive lymphocytes actively participating in this pathological cascade[2].
This article reviews the involvement of B and T cells in atherosclerosis, autoimmune inflammation during atherosclerosis, the autoimmune mechanisms mediated by B cells, T cells and associated cytokines in atherosclerotic plaque development, the immunomodulatory roles of B and T cells in regulating macrophage polarization within atherosclerotic plaques, and the potential value of B and T cells as diagnostic biomarkers for plaque instability in patients with unstable atherosclerosis plaque.
Table of Contents
1. Autoimmune inflammation in atherosclerosis: contributions of B and T cells
2. B cell, T cell, and cytokine-mediated mechanisms in atherosclerotic plaque formation
3. Regulation of macrophage polarization by B and T cells in atherosclerotic lesions
4. Immune and molecular biomarkers of plaque instability in atherosclerosis
01 Autoimmune inflammation in atherosclerosis: contributions of B and T cells
Atherosclerosis is now widely recognized as a chronic inflammatory disorder with a prominent autoimmune component[3]. This disease is characterized by immune responses directed against self-antigens, most notably modified lipoproteins that accumulate within the arterial wall. T cells account for approximately 10% of all cells in human atherosclerotic plaques, of which 70% are CD4+ T cells predominantly of the Th1 phenotype that secretes proatherogenic cytokines including interferon γ (IFN γ) and tumor necrosis factor (TNF)[4]. The presence of oligoclonal T cell expansions in atherosclerotic lesions is indicative of antigen driven immune responses, with a substantial proportion of these T cells recognizing oxidized low density lipoprotein (oxLDL) in an human leucocyte antigen D related(HLA DR) restricted fashion[5],supporting the clinical value of oxLDL assay in evaluating autoimmune related atherogenesis. B cells also contribute to autoimmune inflammation via both antibody dependent and antibody independent mechanisms, and autoantibodies against oxLDL, heat shock proteins (HSPs), and other modified self molecules are detectable in patients with atherosclerosis[2]. Further evidence supporting the autoimmune nature of atherosclerosis comes from the accelerated disease progression observed in patients with systemic lupus erythematosus and rheumatoid arthritis, where traditional risk factors fail to fully explain the increased cardiovascular morbidity[1, 6].
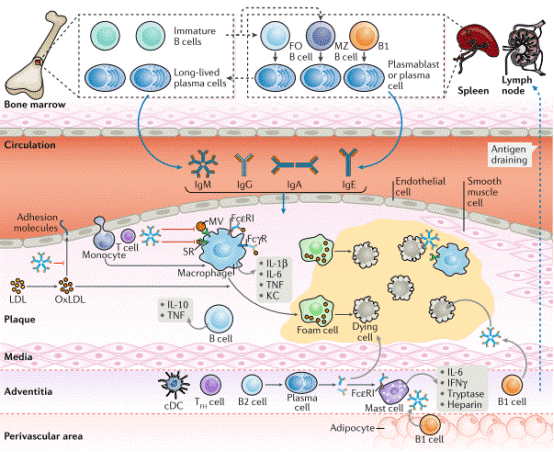
Fig. 1 B cell effector mechanisms in atherosclerotic plaques. B1 cells are mainly of fetal liver origin whereas B2 cells originate from precursors in the bone marrow, which give rise to immature B cells and after maturation in the spleen form follicular (FO) and marginal zone (MZ) B cells. These cells, which mainly home into the bone marrow, secrete immunoglobulins into the blood stream. In the presence of dyslipidaemia, activated endothelium overlying atherosclerotic plaques allows the entry of different immunoglobulins into the plaque area, where they have various functions[7].
02 B cell, T cell, and cytokine-mediated mechanisms in atherosclerotic plaque formation
Atherosclerosis related autoimmune responses are governed by intricate crosstalk among B cells, T cells, and cytokines, which is a key part of b and T cell interaction. CD4+ T cells recognize peptide epitopes derived from apolipoprotein B 100 (ApoB) that are presented by MHC class II molecules on antigen presenting cells. In healthy subjects, the majority of ApoB specific CD4+ T cells exhibit a Foxp3+ regulatory T cell (Treg) phenotype; however, during atherogenic progression, these cells undergo phenotypic switching toward pro inflammatory T helper cell: Th1, Th17, and T follicular helper (Tfh) subsets[8, 9]. B cells exert dual and opposing roles in atherogenesis, either promoting or attenuating disease progression in a subset dependent manner. Specifically, follicular B2 cells accelerate atherosclerosis by driving T cell dependent differentiation into plasma cells that secrete pathogenic IgG antibodies, whereas B1a cells exert atheroprotective effects via the production of natural IgM antibodies targeting oxidation specific epitopes (OSEs)[7, 10]. The CD40-CD40L signaling axis between B and T cells is functionally critical in this context, as B cell specific ablation of either MHCII or CD40 effectively abolishes atherosclerotic lesion development in adoptive transfer models[11]. Cytokines serve as central mediators in coordinating these autoimmune cascades: Th1 derived IFN-γ facilitates macrophage activation and foam cell formation, whereas Interleukin 4 (IL-4) and Interleukin 10 (IL-10) secreted by Th2 and Treg cells mediate anti inflammatory actions, respectively[12]. Interleukin 17 (IL-17) produced by Th17 cells displays context dependent functions, potentially enhancing plaque stability by stimulating collagen synthesis while concurrently contributing to local inflammatory responses[13].
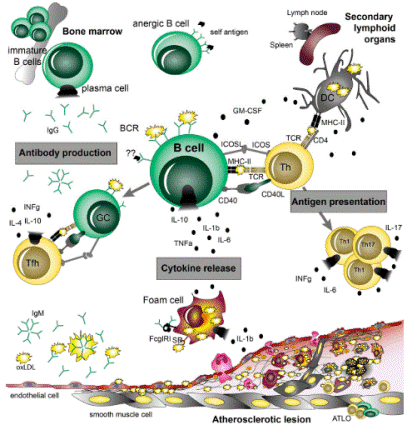
Fig. 2 B cell functions in atherosclerosis. B cells can play both atheroprotective and pro atherogenic roles, which are mediated by their main effector functions: (i) antibody production, (ii) cytokine release, and (iii) antigen presentation interaction with T cells, which are dependent on their subset and activation state[10].
03 Regulation of macrophage polarization by B and T cells in atherosclerotic lesions
Macrophages display substantial plasticity within atherosclerotic lesions, and their polarization status exerts a critical impact on plaque progression and structural stability. M1 M2 macrophage polarization is a key regulatory process here: M1 like pro inflammatory macrophages predominate in unstable, rupture prone plaque regions, whereas M2 like anti inflammatory macrophages are enriched within stable plaque areas and during plaque regression[12]. Tregs directly regulate macrophage function through multiple distinct mechanisms. Tregs suppress the expression of scavenger receptors SRA and CD36 on macrophages, thereby limiting oxLDL internalization and foam cell formation[14]. They also drive macrophage polarization toward an anti inflammatory M2 phenotype characterized by elevated interleukin 10 (IL 10) and transforming growth factor β (TGF β) secretion, accompanied by reduced production of tumor necrosis factor α (TNF α), monocyte chemoattractant protein 1 (MCP 1), and matrixmetalloproteinase 9 (MMP 9). During atherosclerotic regression, Treg derived interleukin 13 (IL 13) stimulates macrophages to synthesize IL 10, thereby enhancing efferocytosis via a Vav1-Rac1 dependent signaling cascade[14]. B cells modulate macrophage polarization through both antibody production and cytokine secretion. Natural IgM antibodies targeting OSEs inhibit macrophage uptake of oxLDL, whereas IgG immune complexes can trigger pro inflammatory macrophage activation through Fc receptor engagement[7]. In addition, Granulocyte macrophage colony stimulating factor (GM CSF) producing IRA B cells exacerbate atherosclerosis by promoting the maturation of conventional dendritic cells and subsequent Th1 cell polarization[10].Recent studies have established that adiponectin mitigates atherosclerotic progression via dual immunometabolic regulatory effects: it promotes macrophage M2 polarization (characterized by increased CD206 and CD163 expression, and decreased CD64 and CD80 levels) and induces T cell exhaustion marked by upregulated expression of programmed cell death protein 1 (PD-1), lymphocyte activation gene 3 protein (LAG3), T cell immunoglobulin and mucin domain-containing protein 3 (TIM3), and cytotoxic T lymphocyte associated protein 4 (CTLA 4), with these effects mediated through inhibition of the NF κB p65/PI3K/Akt signaling pathway[15].
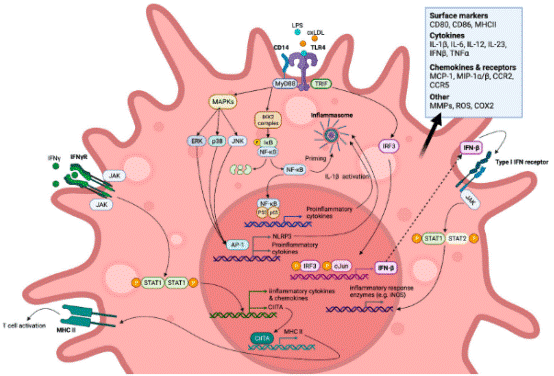
Fig. 3 M1-like polarised macrophage signalling pathways (simplified) induced by toll-like receptor (TLR) and IFN-γ receptor (IFN-γR) endogenous and exogenous agonists[16].
04 Immune and molecular biomarkers of plaque instability in atherosclerosis
Immune cell profiles in the peripheral blood and atherosclerotic plaques correlate with plaque stability and may serve as promising atherosclerosis biomarkers. Compared with stable plaques, unstable plaques present significantly elevated proportions of M0, M1, and M2 macrophages, accompanied by reduced frequencies of CD8+ T cells and NK cells[17]. Machine learning strategies integrating LASSO regression and random forest algorithms have identified M1 macrophages as the most critical cell type underlying plaque instability[17].
4.1 Molecular biomarkers
A number of molecular biomarkers hold potential for the identification of unstable atherosclerotic plaques. CD68 (a pan macrophage marker), PAM (peptidylglycine alpha amidating monooxygenase), and IGFBP6 (insulin like growth factor binding protein 6) exhibit favorable diagnostic performance, with area under the curve (AUC) values of 0.88, 0.90, and 0.86 in training cohorts, respectively, and have been validated in independent datasets and clinical specimens[17]. CD68 displays a positive correlation with plaque instability scores, whereas PAM and IGFBP6 are inversely correlated with plaque instability.
4.2 Inflammatory markers
Circulating levels of inflammatory markers also serve as indicators of plaque vulnerability. Interleukin 6 (IL-6), myeloperoxidase (MPO), and matrix metalloproteinases (most notably MMP-9) are significantly elevated in patients with unstable plaques and can effectively predict cardiovascular events. Pregnancy-associated plasma protein A (PAPP-A) and soluble lectin-like oxidized LDL receptor 1 (sLOX-1) are elevated prior to troponin elevation in acute coronary syndrome, which reflects ongoing plaque inflammation and instability. Additionally, microRNAs (miRNAs), including miR-21, miR-143, miR-223-3p, and miR-122-5p, exhibit differential expression patterns between stable and unstable plaques, thereby providing additional diagnostic potential for identifying plaque vulnerability[18].
4.3 Immune-based biomarkers
Single-cell RNA sequencing (scRNA-seq) analysis of human atherosclerotic carotid plaques has demonstrated that M1 macrophages exhibit the highest "instability score" across all cellular subsets, whereas smooth muscle cells and fibro adipocytes display the highest "stability scores"[17]. These findings underscore the potential of immune-based biomarkers to enhance cardiovascular risk stratification beyond the capabilities of conventional imaging modalities and to inform personalized therapeutic strategies for patients with calcified atherosclerotic disease and atherosclerotic cardiovascular disease.
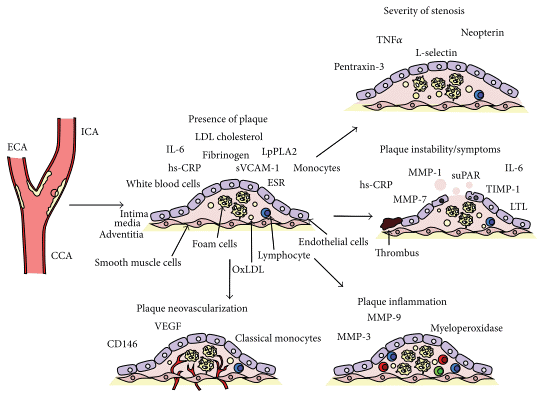
Fig. 4 Summary of the main biomarkers associated with different stages and manifestation of carotid atherosclerosis. CCA: common carotid artery; ICA: internal carotid artery; ECA: external carotid artery; IL: interleukin; LDL: low-density lipoprotein; OxLDL: oxidized lowdensity lipoprotein; ESR: erythrocyte sedimentation rate; hs-CRP: high sensitivity C-reactive protein; sVCAM-1: soluble vascular cell adhesion molecule 1; LpPLA2: lipoprotein-associated phospholipase A2; TNF-α:tumour necrosis factor alpha;MMP: matrixmetalloproteinase; TIMP1: tissue inhibitor of matrix proteinases; suPAR: soluble urokinase plasminogen activator receptor; LTL: leukocyte telomere length; VEGF: vascular endothelialgrowthfactor[19].
In summary, this systematic review delineates the pivotal roles of B and T cells in the autoimmune pathogenesis of atherosclerosis, a chronic inflammatory disorder targeting modified self antigens such as oxLDL. It highlights the dichotomous functions of CD4+ T cell (pro inflammatory Th1/Th17 vs. regulatory Treg) and B cell (pro atherogenic B2 vs. atheroprotective B1a) subsets in plaque inflammation, their immunomodulatory effects on macrophage polarization (Treg promoted M2 phenotype; B cells’ dual effects via antibodies/cytokines), and key biomarkers (e.g., M1 macrophages, CD68, PAM, IGFBP6, miRNAs) for plaque instability and cardiovascular risk stratification. Collectively, these insights advance understanding of autoimmune atherogenesis and provide targets for improving disease management.
Elabscience® Quick Overview of Popular Products
Table 1. Research Tools for atherosclerosis disease
|
Cat. No. |
Product Name |
|
MIH002N |
EasySortTM Human CD4+ T Cell Isolation Kit |
|
MIH008N |
EasySort™ Human Naïve CD8+ T Cell Isolation Kit |
|
MIM005N |
EasySort™ Mouse NK Cell Isolation Kit |
|
XJH001 |
Human Th1/Th2 Flow Cytometry Staining Kit |
|
XJM004 |
RAW 264.7 Polarized M1 Macrophage Induction and Identification Kit |
|
E-HSEL-M0002 |
High Sensitivity Mouse IL-4 (Interleukin 4) ELISA Kit |
|
E-HSEL-H0003 |
High Sensitivity Human IL-6 (Interleukin 6) ELISA Kit |
|
E-HSEL-H0005 |
High Sensitivity Human IL-10 (Interleukin 10) ELISA Kit |
|
E-EL-H0104 |
Human IL-13(Interleukin 13) ELISA Kit |
|
E-CL-H0104 |
Human IL-17 (Interleukin 17) CLIA Kit |
|
E-EL-H2069 |
Human CTLA4(Cytotoxic T-Lymphocyte Associated Antigen 4) ELISA Kit |
|
E-EL-H0081 |
Human GM-CSF(Granulocyte-Macrophage Colony Stimulating Factor) ELISA Kit |
|
E-EL-H0108 |
Human IFN-γ(Interferon Gamma) ELISA Kit |
|
E-EL-H0109 |
Human TNF-α(Tumor Necrosis Factor Alpha) ELISA Kit |
|
E-EL-H1104 |
Human FOXP3(Forkhead Box Protein P3) ELISA Kit |
|
E-EL-H1547 |
Human PD-L1(Programmed Cell Death Protein 1 Ligand 1) ELISA Kit |
|
E-EL-H1587 |
Human TGF-β2(Transforming Growth Factor Beta 2) ELISA Kit |
|
E-EL-H6005 |
Human MCP-1(Monocyte Chemotactic Protein 1) ELISA Kit |
|
E-EL-H6075 |
Human MMP-9(Matrix Metalloproteinase 9) ELISA Kit |
|
E-EL-H6244 |
Human MPO(Myeloperoxidase) ELISA Kit |
|
XJH001 |
Human Th1/Th2 Flow Cytometry Staining Kit |
|
XJM004 |
RAW 264.7 Polarized M1 Macrophage Induction and Identification Kit |
References:
[1] Bartoloni, E., Y. Shoenfeld, and R. Gerli, Inflammatory and autoimmune mechanisms in the induction of atherosclerotic damage in systemic rheumatic diseases: Two faces of the same coin. Arthritis Care & Research, 2011. 63(2): p. 178-183.
[2] Sherer, Y. and Y. Shoenfeld, Mechanisms of Disease: atherosclerosis in autoimmune diseases. Nature Clinical Practice Rheumatology, 2006. 2(2): p. 99-106.
[3] Kobiyama, K. and K. Ley, Atherosclerosis. Circulation Research, 2018. 123(10): p. 1118-1120.
[4] Ketelhuth, D.F.J. and G.K. Hansson, Adaptive Response of T and B Cells in Atherosclerosis. Circulation Research, 2016. 118(4): p. 668-678.
[5] Ammirati, E., et al., The role of T and B cells in human atherosclerosis and atherothrombosis. Clinical and Experimental Immunology, 2015. 179(2): p. 173-187.
[6] Hahn, B.H., et al., The pathogenesis of atherosclerosis in autoimmune rheumatic diseases: Roles of inflammation and dyslipidemia. Journal of Autoimmunity, 2007. 28(2-3): p. 69-75.
[7] Sage, A.P., et al., The role of B cells in atherosclerosis. Nature Reviews Cardiology, 2018. 16(3): p. 180-196.
[8] Saigusa, R., H. Winkels, and K. Ley, T cell subsets and functions in atherosclerosis. Nature Reviews Cardiology, 2020. 17(7): p. 387-401.
[9] Ley, K., Role of the adaptive immune system in atherosclerosis. Biochemical Society Transactions, 2020. 48(5): p. 2273-2281.
[10] Shelby D. Ma, M.M., Functional Role of B Cells in Atherosclerosis. Cells, 2021.10(2): 270
[11] Tay, C., et al., B Cell and CD4 T Cell Interactions Promote Development of Atherosclerosis. Frontiers in Immunology, 2020. 10:3046.
[12] Tabas, I. and A.H. Lichtman, Monocyte-Macrophages and T Cells in Atherosclerosis. Immunity, 2017. 47(4): p. 621-634.
[13] Bäck, M., C. Weber, and E. Lutgens, Regulation of atherosclerotic plaque inflammation. Journal of Internal Medicine, 2015. 278(5): p. 462-482.
[14] Ouyang, X. and Z. Liu, Regulatory T cells and macrophages in atherosclerosis: from mechanisms to clinical significance. Frontiers in Immunology, 2024. 15:1435021.
[15] Qin, M., et al., Adiponectin attenuates atherosclerosis via macrophage polarization-mediated T Cell exhaustion by modulating the NF-κB p65/PI3K/Akt signaling pathway. Tissue and Cell, 2025:103150.
[16] Sheridan, A., C.P.D. Wheeler-Jones, and M.C. Gage, The Immunomodulatory Effects of Statins on Macrophages. Immuno, 2022. 2(2): p. 317-343.
[17] Wang, J., et al., Identification of immune cell infiltration and diagnostic biomarkers in unstable atherosclerotic plaques by integrated bioinformatics analysis and machine learning. Frontiers in Immunology, 2022. 13:956078.
[18] Lubrano, V. and S. Balzan, Status of biomarkers for the identification of stable or vulnerable plaques in atherosclerosis. Clinical Science, 2021. 135(16): p. 1981-1997.
[19] Ammirati, E., et al., Markers of Inflammation Associated with Plaque Progression and Instability in Patients with Carotid Atherosclerosis. Mediators of Inflammation, 2015. 2015(1):718329.



