The in vitro activation, culture, and expansion of T cells represent a core process in cellular immunotherapy, with outcomes directly impacting the safety and efficacy of treatment. Our previous post provided a preliminary exploration of the differences in expansion capacity and phenotypic changes among various starting cell populations through a 20-day dynamic expansion comparison of human PBMCs, sorted CD3+ T cells, and Pan Naive T cells. How to further enhance T cell expansion efficiency, maintain their functional status, and delay exhaustion constitutes a critical challenge in current T cell research and application.
This article will systematically review the main factors influencing the in vitro T cell expansion process, including cytokine combinations, activation and stimulation methods, culture medium nutrients, co-stimulatory molecule systems, and other key influencing factors. It will introduce the essential technical details of long-term T cell culture to provide references for optimizing cell therapy protocols.
Table of Contents
1. Cytokine-driven regulation of T cell expansion, differentiation, and functional maintenance
2. Optimization of T cell activation strategies: antibody-vs APC-mediated stimulation
3. Metabolic and nutritional control of T cell proliferation and long-term culture stability
4. Co-stimulatory signaling networks in T cell activation, persistence, and exhaustion control
5. Additional determinants of T cell expansion: cell source, subsets, and functional state
6. Case study: 16-color spectral flow cytometry profiling of human T cell subsets
01 Cytokine-driven regulation of T cell expansion, differentiation, and functional maintenance
The in vitro culture and expansion of T cells are inseparable from the support of cytokines, among which the γc (common gamma chain) family of cytokines (including IL-2, IL-7, IL-15, IL-21, etc.) are the most important class. These cytokines regulate T cell proliferation, differentiation, survival, and function by binding to specific receptors on the T cell surface and activating downstream signaling pathways.
Table 1. The Impact of the Cytokine System on T Cells
|
Cytokine Type |
Optimal Concentration |
Receptor Type |
Impact Mechanism |
Effect |
Feature |
|
IL-2 |
50-200 IU/mL |
IL-2R (α, β, γ chains) |
Binds to the high-affinity IL-2 receptor, activating downstream signaling pathways and driving cell cycle progression. |
Promotes T cell entry from the G0 phase into the cell cycle, driving robust proliferation. |
Dual nature: It both expands effector T cells and stimulates the growth of regulatory T cells (Tregs), potentially suppressing anti-tumor immunity. |
|
IL-7 |
5-10 ng/mL |
IL-7R |
Activates anti-apoptotic genes and suppresses the expression of pro-apoptotic genes. |
It is crucial for the survival of naive and memory T cells, prolonging their lifespan and maintaining homeostasis. |
Advantage: Does not stimulate Tregs or NK cells. When used alone, it does not induce significant proliferation. |
|
IL-15 |
2.5-10 ng/mL |
IL-15 (Rα, β, γ chains) |
Primarily functions through "trans-presentation" (where IL-15 binds to IL-15Rα and is then presented to neighboring T cells). |
Efficiently promotes CD8+ T cell proliferation and memory formation; maintains an "effector memory phenotype." |
Advantage: Enhances cytotoxicity and avoids exhaustion caused by over-activation, with low expression of exhaustion markers. |
|
IL-21 |
10-20 ng/mL |
IL-21R |
Enhances T cell effector functions through signaling pathways such as STAT3. |
Enhances T cell killing function (promotes granzyme and IFN-γ secretion); induces the formation of central memory-like T cells. |
Advantage: Reduces the expression of exhaustion markers such as PD-1, focusing on enhancing effector function rather than promoting proliferation. |
In T cell culture, the function of a single cytokine is limited, so combination strategies are often employed to achieve synergistic effects, resulting in a "1+1>2" outcome. Studies have shown that different cytokine combinations have distinct focuses:
(1) The combination of IL-2 and IL-7 is the most classic foundational protocol. By using IL-2 to promote rapid proliferation and IL-7 to maintain cell survival, it can preserve cell function while ensuring expansion efficiency.
(2) The combination of IL-7 and IL-15 focuses more on maintaining the memory phenotype and stem cell-like properties of T cells, reducing terminal differentiation, thereby enhancing the in vivo persistence and anti-tumor efficacy of cells like CAR-T.
(3) A triple combination including IL-2, IL-7, and IL-15 provides the most comprehensive support, achieving expansion folds of over 255 times, making it particularly suitable for scenarios requiring long-term culture.
Furthermore, the addition of IL-21 can synergistically enhance proliferation, antagonize regulatory T cell development, and boost killing activity. Other factors like IFN-γ and TNF-α can also be used in combination with γc family cytokines to further optimize culture outcomes by enhancing antigen presentation or providing auxiliary signals.
02 Optimization of T cell activation strategies: antibody- vs APC-mediated stimulation
The in vitro activation of T cells is a critical step in CAR-T preparation and is primarily divided into two strategies: antibody-mediated activation and antigen-presenting cell (APC)-mediated activation.
➢ Antibody-mediated activation most commonly uses the anti-CD3/CD28 bead system. It offers strong signaling, high expansion efficiency, and easy separation. The bead-to-cell ratio significantly affects the activation outcome. Meanwhile, novel strategies such as lipid microbubbles, antibody fragments, and bispecific antibodies are continuously being optimized.
➢ Natural APC activation more closely mimics the physiological state, but it has limitations such as significant individual variability and complex preparation. Artificial APCs (aAPCs) allow for standardization and precise control, offering greater advantages in industrial applications.
Application: Different activation methods possess distinct characteristics in terms of expansion efficiency, specificity, cost, signal persistence, and clinical suitability. Antibody activation is often used for large-scale T cell expansion, while APC/aAPC methods are more suitable for generating antigen-specific T cells. A rational choice should be made based on specific research and clinical requirements.
03 Metabolic and nutritional control of T cell proliferation and long-term culture stability
Nutritional components in the culture environment are crucial for regulating T cell proliferation and function in vitro, primarily involving amino acids, vitamins, energy substrates, and optimization of the culture medium system.
Amino Acids: Amino acids such as glutamine, arginine, and cysteine participate in energy metabolism, antioxidant defense, and regulation of the mTOR pathway, influencing T cell proliferation and exhaustion.
Vitamins: B vitamins, vitamin C, and vitamin E help maintain metabolism, support stem cell-like phenotypes, and enhance antioxidant capacity, respectively.
Energy Substrates: Glucose is the core energy substrate, and upon T cell activation, glucose metabolism shifts towards glycolysis. Glucose concentrations must be maintained within an appropriate range. Glutamine, fatty acids, and ketone bodies can serve as supplementary energy sources, with fatty acid oxidation being particularly important for the maintenance of memory T cells.
Culture Medium Systems: Regarding the culture medium itself, traditional serum-containing formulations have issues with batch-to-batch variability and safety risks. Serum-free, chemically defined media enhance stability and safety. When combined with additives like N-acetylcysteine (NAC), inositol, and appropriate control of pH and osmolality, these media can significantly improve T cell expansion efficiency and functional quality, meeting the requirements for clinical-grade cell manufacturing.
04 Co-stimulatory signaling networks in T cell activation, persistence, and exhaustion control
Co-stimulatory molecules are core elements that regulate T cell activation, differentiation, and function, primarily including the CD28 family and the TNF receptor superfamily (such as 4-1BB, OX40, CD27, GITR, etc.). Different co-stimulatory molecules possess distinct regulatory functions.
➢ CD28 provides the essential second signal for T cell activation, influencing proliferation and effector function.
➢ 4-1BB focuses on maintaining memory T cells, enhancing survival, and improving mitochondrial function.
➢ OX40 and CD27 can synergistically promote the memory phenotype and anti-tumor activity.
Single co-stimulatory molecules have limitations, whereas various combinations can exert synergistic effects. Among these, the combination of CD28 and 4-1BB is the most classic strategy used in CAR-T therapy, balancing rapid expansion with long-term persistence. The absence of co-stimulatory signals can exacerbate T cell exhaustion. Conversely, the rational application of co-stimulatory molecules like 4-1BB or combined signals can rescue exhausted T cells, improve metabolism and function. Integrating novel bispecific antibodies and immune checkpoint blockade strategies can further enhance T cell persistence and anti-tumor efficacy. Furthermore, multi-combination co-stimulation can directionally shape specific functional T cell subsets.
05 Additional determinants of T cell expansion: cell source, subsets, and functional state
In addition to the cytokine combinations, activation and stimulation methods, culture medium nutrients, and co-stimulatory molecule systems discussed above, other major factors influencing the in vitro T cell expansion process include the source and initial state of the T cells. The source and initial state of T cells are important factors affecting culture outcomes, with T cells from different sources and states exhibiting significant differences during culture.
T Cell Source: T cells can be obtained from various tissues, each with distinct characteristics. Examples include peripheral blood (the most common source, yielding peripheral blood mononuclear cells containing 50%-70% T cells), umbilical cord blood (more primitive cells with strong proliferative capacity and low immunogenicity), tumor-infiltrating lymphocytes (TILs, which are tumor-specific but complex to isolate and culture, with high individual variability), and lymph nodes.
T Cell Subset Culture: CD8+ T cells are more sensitive to IL-2 and expand more readily, while CD4+ T cells proliferate more slowly but are important for maintaining culture balance. The proportion of CD8+ T cells typically increases during culture.
T Cell State: Naive T cells require stronger activation signals but possess greater proliferation potential once activated. In contrast, memory T cells are more easily activated but have limited proliferation potential and are prone to exhaustion during long-term culture.
Other Factors: Furthermore, patient-specific factors such as age, disease status, and treatment history can influence T cell culture outcomes. T cells derived from elderly individuals or cancer patients often show reduced proliferation and increased exhaustion.
06 Case study: 16-color spectral flow cytometry profiling of human T cell subsets
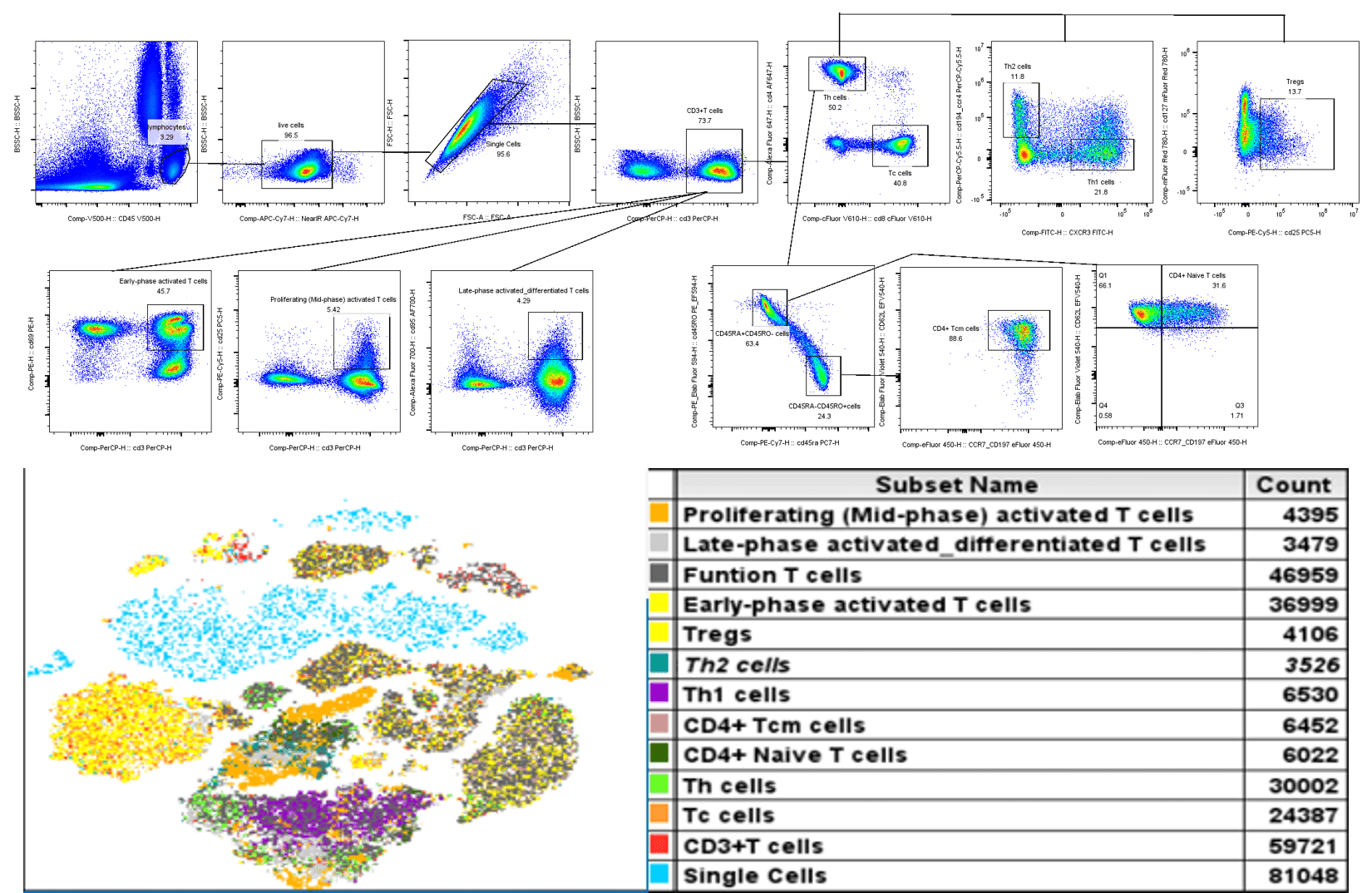
Fig. 1 16-color full spectrum analysis of human peripheral blood T cells. T cell subsets in human peripheral blood cells were detected using a spectral flow cytometer (Agilent Novocyte Opteon VBRY), and the results were analyzed using FLOWJO software.
Relevant products:
Table 2. Antibody Panel for 16-Color Spectral Flow Cytometry of Human Peripheral Blood T Cell Subsets
|
Marker |
Clone. No |
Fluorochrome |
Cat. No. |
|
OKT-8 |
Elab Fluor® Violet 610 |
E-AB-F1110T |
|
|
G043H7 |
Elab Fluor® Violet 450 |
E-AB-F1159Q |
|
|
OKT-3 |
Percp |
E-AB-F1001F |
|
|
SK3 |
Elab Fluor® 647 |
E-AB-F1352M |
|
|
HI100 |
PE/Cyanine7 |
E-AB-F1052H |
|
|
HI30 |
Elab Fluor® Violet 500 |
E-AB-F1137R |
|
|
CD28.2 |
APC |
E-AB-F1195E |
|
|
FN50 |
PE |
E-AB-F1138D |
|
|
UCHL1 |
PE/Elab Fluor® 594 |
E-AB-F1139P |
|
|
DX2 |
Elab Fluor® 700 |
E-AB-F1168M1 |
|
|
BC96 |
PE/Cyanine5 |
E-AB-F1194G |
|
|
A019D5 |
Elab Fluor® Red 780 |
E-AB-F1152S |
|
|
DREG56 |
Elab Fluor® Violet 540 |
E-AB-F1051T3 |
|
|
G025H7 |
FITC |
E-AB-F1156C |
|
|
L291H4 |
PerCP/Cyanine5.5 |
E-AB-F1366J |
|
|
/ |
STYX™ Near-IR |
E-CK-A168 |
References:
[1] Hashimoto M, Im SJ, Araki K, Ahmed R. Cytokine-Mediated Regulation of CD8 T-Cell Responses During Acute and Chronic Viral Infection. Cold Spring Harb Perspect Biol. 2019 Jan 2;11(1):a028464. doi: 10.1101/cshperspect.a028464.
[2] Sprent J, Boyman O. Optimising IL-2 for Cancer Immunotherapy. Immune Netw. 2024 Jan 26;24(1):e5. doi: 10.4110/in.2024.24.e5.
[3] Dong C. Cytokine Regulation and Function in T Cells. Annu Rev Immunol. 2021 Apr 26;39:51-76. doi: 10.1146/annurev-immunol-061020-053702.
[4] Bell M, Gottschalk S. Engineered Cytokine Signaling to Improve CAR T Cell Effector Function. Front Immunol. 2021 Jun 4;12:684642. doi: 10.3389/fimmu.2021.684642.
[5] Pietrobon V, Todd LA, Goswami A, Stefanson O, Yang Z, Marincola F. Improving CAR T-Cell Persistence. Int J Mol Sci. 2021 Oct 7;22(19):10828. doi: 10.3390/ijms221910828.
[6] Deng S, Sun Z, Qiao J, Liang Y, Liu L, Dong C, Shen A, Wang Y, Tang H, Fu YX, Peng H. Targeting tumors with IL-21 reshapes the tumor microenvironment by proliferating PD-1intTim-3-CD8+ T cells. JCI Insight. 2020 Apr 9;5(7):e132000. doi: 10.1172/jci.insight.132000.
[7] Tovar Manzano D, Subhi-Issa N, Pereiro-Rodríguez A, López Cade IG, Mateos González M, Fernández Arquero M, Pérez Segura P, Ujaldón Miró C, Sánchez-Ramón S, Ocaña A, Guzmán Fulgencio M. Tumor-infiltrating lymphocyte expansion protocols for adoptive cell therapy in cancer. Cell Oncol (Dordr). 2025 Dec;48(6):1659-1675. doi: 10.1007/s13402-025-01112-2. Epub 2025 Nov 17.
[8] Ghaffari S, Torabi-Rahvar M, Aghayan S, Jabbarpour Z, Moradzadeh K, Omidkhoda A, Ahmadbeigi N. Optimizing interleukin-2 concentration, seeding density and bead-to-cell ratio of T-cell expansion for adoptive immunotherapy. BMC Immunol. 2021 Jul 3;22(1):43. doi: 10.1186/s12865-021-00435-7.
[9] Boustani J, Joseph ELM, Martin E, Benhmida S, Lecoester B, Tochet F, Mirjolet C, Chevalier C, Thibouw D, Vulquin N, Servagi S, Sun X, Adotévi O. Cisplatin-based chemoradiation decreases telomerase-specific CD4 TH1 response but increases immune suppressive cells in peripheral blood. BMC Immunol. 2021 Jun 18;22(1):38. doi: 10.1186/s12865-021-00429-5.
[10] Pan C, Li J, Chen S, Yu H, Ye Y. [IL-7/IL-15/IL-21/IL-23 effectively promote the generation of human CD8+ central memory T cells in vitro]. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2021 Oct;37(10):872-880. Chinese. PMID: 34670663.
[11] Dong J, Yang SX, Li Y, Gao JP, Zhang X. [In vitro expansion of T cells stimulated by combination of IL-2, IL-7 and IL-15]. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2010 Dec;18(6):1590-4. Chinese. PMID: 21176376.
[12] Abdelsamed HA, Zebley CC, Youngblood B. Epigenetic Maintenance of Acquired Gene Expression Programs during Memory CD8 T Cell Homeostasis. Front Immunol. 2018 Jan 18;9:6. doi: 10.3389/fimmu.2018.00006.



