CAR-T cell therapy has demonstrated remarkable efficacy in treating hematologic malignancies but continues to face challenges in solid tumor treatment, partly due to the need for improved T-cell expansion efficiency, phenotypic stability during in vitro culture, and in vivo persistence. In the process of T-cell in vitro culture, optimizing T-cell purity and phenotypic stability through negative sorting and precise activation is a critical step in the field of cell therapy, particularly for the success of chimeric antigen receptor T-cell (CAR-T) therapy.
Table of Contents
1. Negative sorting and precise activation for T cell culture optimization
2. Common problems and solutions in negative isolation and T cell activation
01 Negative sorting and precise activation for T cell culture optimization
T cell sorting and activation, as the fundamental steps in in vitro culture, often encounter three major challenges in practical operation: Traditional positive sorting is prone to leaving residual exogenous magnetic beads and altering the conformation of the receptor through antibody binding, leading to non-specific activation and phenotypic drift; Fixed-intensity activation protocols are likely to cause excessive stimulation of T cells, premature exhaustion of cells, and loss of memory phenotype; and a disjointed between the isolation and activation steps can lead to a sharp decline in cell viability due to prolonged operation time.
To address these issues, the strategy of negative sorting combined with precise activation can be adopted to achieve controlled stimulation. This is a key approach for controlling the quality of T cells at the source and optimizing the subsequent cultivation results, as shown in the table below:
Table 1. Combined Strategy of Negative Sorting + Precise Activation
|
Strategy |
Principle |
Advantages |
Key Points |
|
Negative sorting (CD3+ T cells or Pan Naive T cells) |
By labeling non-T cells (such as B cells and monocytes) in the sample with specific antibodies, and then using magnetic beads to remove non-target cells, the unmarked target T cells can be indirectly enriched. |
1) No exogenous markers or magnetic bead residues, avoiding changes in the conformation of T cell surface receptors; 2) The target T cells are not subjected to additional stimulation, and the live cell rate can reach over 95%; 3) The phenotype is close to the natural state in the body, suitable for all subsequent experiments (activation, transfection, long-term culture, etc.); 4) Separation is highly efficient, taking only 20 minutes for the entire process. |
1) Choose the CD3+ T cell negative isolation kit, suitable for PBMCs, mouse splenocytes/lymph node cells; 2) Incubate with antibodies for 5 min at room temperature, then incubate with magnetic beads for 5 min; mix gently; 3) Perform magnetic separation by standing for 5 min at room temperature; collect only the supernatant (avoid touching the bead complex); 4) Post-isolation flow cytometry detection shows purity ≥95.98% and viable cell rate ≥98%; Note: Cell yield is higher with fresh samples; please use fresh samples whenever possible. |
|
Precise Activation |
Establish a controllable system for efficient stimulation of linked cytokine expansion culture by anti-CD3/CD28 Beads, following the principle of "moderate activation to initiate proliferation, precise regulation to maintain phenotype", and avoiding the damage to cells caused by traditional strong stimulation. |
1) Prevent premature exhaustion of T cells due to excessive activation; 2) Effectively maintain the memory phenotype (TN/TCM) and reduce the expression of exhaustion markers (PD-1, LAG3); 3) Stable activation efficiency, suitable for subsequent long-term cultivation and cell therapy research and development needs. |
1) Basic stimulation: Mix CD3/CD28 Beads with CD3+ T cells in a suitable ratio, activate for 3-5 days, then remove the CD3/CD28 Beads using magnetic suction. Magnetic separation can be performed twice to ensure thorough removal of the activation Beads; 2) Cytokine expansion culture: Initially culture the cells with a low dose of IL-2 (50 U/mL). After 24 hours, add IL-7 and IL-15 (5 ng/mL each). Adjust the cell passage density to 5×10⁵/mL, refresh the medium every 48 h, and simultaneously supplement with cytokines. |
Note: The two strategies need to be seamlessly connected. Try to use fresh samples for isolation, and quickly add CD3/CD28 Beads within 30-60 minutes after isolation to establish the activation system. Throughout the process, minimize cell exposure to room temperature and excessive pipetting to maintain cell viability and phenotypic stability. Additionally, excessively high IL-2 levels can lead to T cell exhaustion; IL-2 may not be added during the initial activation phase, and the IL-2 concentration should not exceed 50 U/mL during post-activation culture. If overactivation leads to T cell exhaustion, reduce the activation time or decrease the ratio of CD3/CD28 beads.
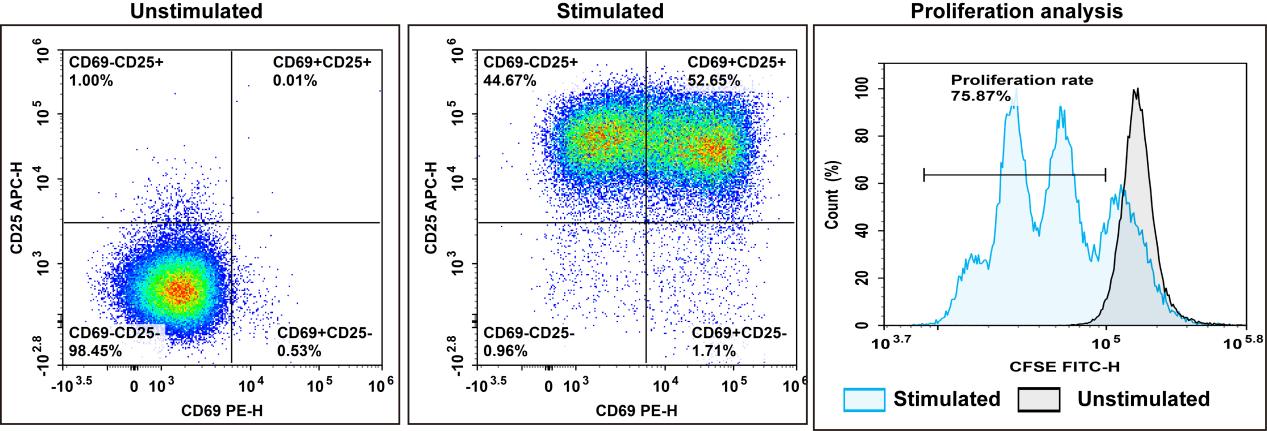
Fig. 1 Experimental results of negative isolation and rapid activation of human CD3+ T cells. After isolating PBMCs from human peripheral blood using Human PBMC Separation Solution (P1.077) (E-CK-A103), CD3+ T cells with a purity of 98.2% were isolated using the EasySort™ Human CD3+ T Cell Isolation Kit (MIH001N). Following staining with the CFSE Cell Division Tracker Kit (E-CK-A345), the cells were activated with Human CD3/CD28 T Cell Activation Beads (MIH001A) and cultured for 72 h, after which cell activation efficiency was assessed.
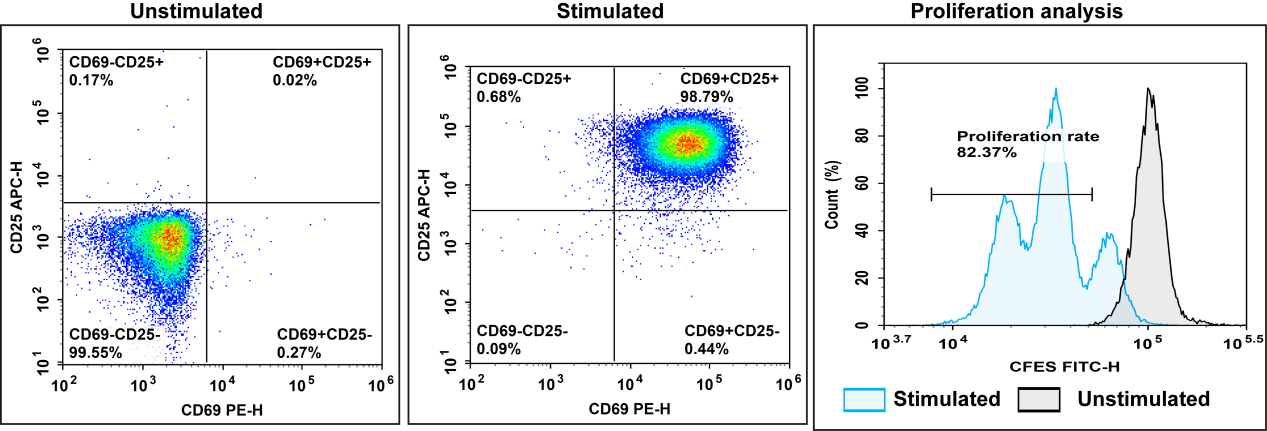
Fig. 2 Experimental results of negative isolation and rapid activation of human Naïve Pan T cells. After isolating PBMCs from human peripheral blood using Human PBMC Separation Solution (P1.077) (E-CK-A103), Naïve Pan T cells with a purity of 97.4% were isolated using the EasySort™ Human Naïve Pan T Cell Isolation Kit (MIH006N). Following staining with the CFSE Cell Division Tracker Kit (E-CK-A345), the cells were activated with Human CD3/CD28 T Cell Activation Beads (MIH001A) and cultured for 72 hours, after which cell activation efficiency was assessed.
02 Common problems and solutions in negative isolation and T cell activation
The combined strategy of negative isolation and precise activation involves a complex experimental workflow with numerous detailed operations. Common problems, possible reasons, and solutions are shown in the table.
Table 2. Common problems in Practical Operations and Their Solutions
|
Common Problem |
Possible Reason |
Solution |
|
After negative sorting, the purity of CD3+ cells was less than 90% |
1) Dead cells not removed; 2) Insufficient antibody/bead incubation time; 3) Inadequate magnetic separation. |
1) Process the samples with the dead cell removal kit in advance; 2 According to the product manual, appropriately extend the incubation time (antibody 20 minutes, magnetic beads 10 minutes); 3) If the magnetic stirrer is not compatible or the magnetic force is insufficient, appropriately increase the static time for magnetic separation, and repeat the magnetic separation once. |
|
After activation, the cell viability is less than 80% |
1) The time between sorting and activation exceeds 30 minutes; 2) The culture medium/reagents are not pre-warmed; 3) The initial cell density is inappropriate. |
1) Use fresh samples and operate strictly according to the instructions. Complete activation within 30 minutes and avoid excessive cell shaking; 2) All reagents should be pre-warmed at 37℃; 3) Adjust the density to 5×105/mL. Using ultra-low adsorption culture plates can reduce non-specific adsorption of cells during the culture process. Reduce half of the medium after 24 hours, and replace the entire medium and add cytokines at 48 hours to maintain the concentration of nutrients and cytokines in the culture medium and ensure the continuous expansion of T cells. |
|
After activation, phenotypic drift occurs, and the proportion of memory phenotypes significantly decreases |
1) The ratio of CD3/CD28 Beads is too high or the activation time is too long; 2) IL-7 and IL-15 are not added; 3) The dose of IL-2 is too high in the initial stage of cultivation. |
1) Appropriately reduce the proportion of CD3/CD28 Beads, and shorten the activation time to 3 hours. The maximum duration should not exceed 5 days. 2) Appropriately add the combination factor of IL-7 and IL-15. 3) Keep the concentration of IL-2 within 50 U/mL. Excessive dosage may cause cell exhaustion. |
|
After activation, the cells proliferated slowly without obvious clumping |
1) Insufficient stimulation intensity or excessive stimulation; 2) Lack of co-stimulatory molecules; 3) Abnormal culture conditions. |
1) Adjust the appropriate ratio of CD3/CD28 Beads and the activation time. Under the microscope, clear cell clusters can be observed. 2) Add the co-stimulatory molecules CD27/CD137 (4-1BB) and the cytokines IL-7/IL-15/IL-2. 3) Check the incubator and avoid overly tight sealing of the culture plate to prevent hypoxia. |
The key to in vitro cultivation of T cells lies in obtaining T cells with high purity, stable phenotype and excellent activity. The strategy of negative sorting combined with precise activation has fundamentally solved core issues such as exogenous residues and unstable phenotype. Additionally, by coordinating with standardized operation procedures, meticulous process control and systematic problem identification, high-quality T cells can be obtained.
References:
[1] Deckers, J., Anbergen, T., Hokke, A. M., de Dreu, A., Schrijver, D. P., de Bruin, K., Toner, Y. C., Beldman, T. J., Spangler, J. B., de Greef, T. F. A., Grisoni, F., van der Meel, R., Joosten, L. A. B., Merkx, M., Netea, M. G., & Mulder, W. J. M. (2023). Engineering cytokine therapeutics. Nature Reviews Bioengineering, 1(4), 286–303. https://doi.org/10.1038/s44222-023-00030-y.
[2] Chan, J. D., Lai, J., Slaney, C. Y., Kallies, A., Beavis, P. A., & Darcy, P. K. (2021). Cellular networks controlling T cell persistence in adoptive cell therapy. Nature Reviews Immunology, 21(12), 769–784. https://doi.org/10.1038/s41577-021-00539-6.



