Interleukins (ILs) are key cytokine messengers that regulate immune cell communication, inflammation, and tissue homeostasis. This review focuses on the functional roles of Interleukin-1 (IL-1), Interleukin-6 (IL-6), Interleukin-7 (IL-7), and Interleukin-8 (IL-8) in health and disease, with a focus on their interleukins function and interleukin signaling. IL-1 and IL-6 serve as central pro-inflammatory mediators bridging innate and adaptive immunity; IL-7 is essential for lymphocyte development and survival; and IL-8 directs neutrophil chemotaxis. The balanced activity of these interleukins is critical, as their dysregulation contributes to autoinflammatory diseases, chronic inflammation, and cancer, which are key aspects of interleukins in disease. Herein, we discuss their signaling mechanisms, immune-modulatory effects, and relevance as therapeutic targets.
This article reviews the overview of interleukins and their general functions, the activation pathways and immune regulation of IL-1, the pro-inflammatory signaling and disease-related role of IL-6, the regulatory effects of IL-7 on lymphocyte development and homeostasis, the chemotactic function and neutrophil recruitment mediated by IL-8, the measurement of interleukin levels using ELISA and AptplexTM assays, the crosstalk between interleukins and macrophage polarization, as well as the interleukin-mediated modulation of the Treg/Th17 balance.
Table of Contents
1. Overview of interleukins and their general functions
2. IL-1: activation pathways and immune regulation
3. IL-6: pro-inflammatory signaling and role in disease
4. IL-7: regulation of lymphocyte development and homeostasis
5. IL-8: chemotactic function and neutrophil recruitment
6. Measurement of IL levels using ELISA and AptplexTM assays
7. Crosstalk between ILs and macrophage polarization
8. IL-mediated modulation of Treg/Th17 balance
01 Overview of interleukins and their general functions
According to the attached literature, ILs are low-molecular-weight proteins primarily produced by leukocytes to mediate communication among white blood cells. They are widely involved in cell growth, differentiation, inflammation, immunity, and tissue repair[1]. IL-1 (existing as Interleukin-1α and Interleukin-1β) and IL-6, produced by macrophages, endothelial cells, and other cell types,promote T and B cell activation, induce fever, regulate acute-phase responses, and contribute to inflammatory processes. Interleukin-2 (IL-2), primarily secreted by T cells, serves as a key growth factor for T cells, natural killer (NK) cells, and lymphokine-activated killer (LAK) cells. Interleukin-3 (IL-3) supports the survival and differentiation of multipotent hematopoietic progenitors. Interleukin-4 (IL-4) not only promotes B cell proliferation and immunoglobulin (Ig)E/IgG1 production but also modulates T cell and mast cell functions. Interleukin-5 (IL-5) specifically drives the growth and activation of eosinophils[2]. IL-7, produced by bone marrow stromal cells and thymic cells, mainly supports the growth of B and T lymphocyte precursors. IL-8 (also known as neutrophil-activating peptide 1, NAP-1), a member of the α-chemokine family, is a potent chemoattractant for neutrophils and can also induce lysosomal enzyme release and enhance neutrophil adhesion. These interleukins exert their effects through their respective high-affinity receptors, exhibiting both specific and overlapping functions, and together form a complex immune network that is fundamental to understanding their roles in health and disease.
02 IL-1: activation pathways and immune regulation
The IL-1 family (especially IL-1β and IL-1α) is a core initiator of inflammatory responses and serves as a bridge between innate and adaptive immunity[3]. The maturation and secretion of IL-1β are tightly regulated by a "two-signal" mechanism: the first signal (e.g., lipopolysaccharide (LPS) and other toll-like receptor (TLR) ligands) induces pro-IL-1β synthesis; the second signal (e.g., extracellular ATP, uric acid crystals, cholesterol crystals) activates the NLRP3 inflammasome, which in turn activates caspase-1 to cleave pro-IL-1β into biologically active mature IL-1β[3-5]. The detection of mature IL-1β can be achieved through IL-1β elisa, a reliable method for quantifying this cytokine. Unlike IL-1β, the IL-1α precursor is intrinsically active and, upon cell necrosis, is released as an "alarmin" that directly activates IL-1R1 signaling on neighboring cells, mediating sterile inflammation[6, 7]. At the signal transduction level, IL-1 binding to IL-1R1 recruits the co-receptor IL-1R3 (IL-1RAcP) and activates the MyD88-dependent pathway, sequentially engaging IRAK family members, TRAF6, and TAK1, ultimately triggering the nuclear factor-κB (NF-κB) and mitogen-activated protein kinase (MAPK; JNK/p38) pathways and inducing the expression of hundreds of inflammation-related genes, including IL-6, IL-8, and cyclooxygenase-2 (COX-2)[5, 6]. The IL-1 system is subject to multiple negative feedback mechanisms, including IL-1Ra (receptor antagonist), IL-1R2 (decoy receptor), IL-1R8 (SIGIRR), and the anti-inflammatory cytokine Interleukin-37 (IL-37), which together prevent excessive inflammation[4, 6]. In terms of immune regulation, IL-1 is not only a key co-stimulatory factor for the differentiation and maintenance of T helper 17 (Th17) cells but also synergizes with Interleukin-23 (IL-23) to enhance Interleukin-17 (IL-17) production, and it participates in the activation of T helper 1 (Th1), T helper 2 (Th2), and CD8+ T cells[4, 7]. Moreover, IL-1 drives emergency hematopoiesis and trained immunity, thereby long-term reshaping the epigenetic and metabolic status of innate immune cells. Based on these mechanisms, IL-1 has become an important therapeutic target in various autoinflammatory diseases, atherosclerosis, and cancer; IL-1Ra (anakinra) and anti-IL-1β antibodies (canakinumab) have shown significant clinical efficacy. A thorough understanding of the IL-1 activation pathways and its immunoregulatory networks is of great importance for designing in vitro cell models, establishing animal models of inflammatory diseases, and developing novel anti-inflammatory strategies.
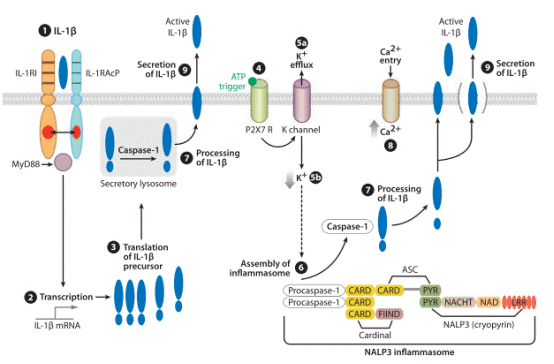
Fig. 1 Steps in the synthesis and secretion of IL-1β induced by IL-1β. Regardless of the stimulus, in monocytes and macrophages specific inhibitors of caspase-1 reduce the secretion of mature IL-1β, and precursor IL-1β accumulates mostly inside the cell. The rate-limiting step in the processing and secretion of IL-1β takes place with activation ofthe inflammasome[3].
03 IL-6: pro-inflammatory signaling and role in disease
Interleukin-6 (IL-6) is a pleiotropic cytokine initially discovered for its ability to induce B lymphocytes to produce immunoglobulin, and thus has been regarded as an important pro-inflammatory factor. The quantification of IL-6 levels is commonly performed using IL-6 elisa, with IL-6 elisa kit being a widely used tool for this purpose. IL-6 is mainly produced by monocytes/macrophages, T cells, fibroblasts, and various tissue-resident cells; its expression is rapidly upregulated upon infection, injury, or stimulation by pro-inflammatory cytokines such as IL-1β and tumor necrosis factor-α (TNF-α)[8]. IL-6 signaling is primarily mediated through two mechanisms: classic signaling (via the membrane-bound IL-6 receptor α subunit) and trans-signaling (via the soluble IL-6 receptor)[8, 9]. In classic signaling, only cells expressing the membrane-bound IL-6R (e.g., hepatocytes, some leukocytes) can respond to IL-6; in contrast, trans-signaling allows the IL-6/sIL-6R complex to activate all gp130-expressing cells, greatly broadening its range of action[10]. Accumulating evidence indicates that IL-6 trans-signaling is critical for driving chronic inflammation and autoimmune diseases, whereas classic signaling primarily participates in anti-inflammatory, tissue-regenerative, and acute-phase responses[9, 11].
Regarding diseases, dysregulated IL-6 is closely associated with various autoimmune and inflammatory conditions. Elevated IL-6 levels are detected in the serum or local tissues of patients with rheumatoid arthritis, systemic lupus erythematosus, and IgA nephropathy, and correlate positively with disease activity[8, 12]. IL-6 also promotes the differentiation of CD4+ T cells into Th17 cells while inhibiting regulatory T cell (Treg) function, thereby breaking immune tolerance and exacerbating tissue damage[12]. Furthermore, IL-6 exerts pro-tumorigenic effects in inflammation-associated cancers (e.g., colon cancer, pancreatic cancer) by activating the signal transducer and activator of transcription 3 (STAT3) signaling pathway to promote tumor cell proliferation, survival, and angiogenesis. Clinically, monoclonal antibodies targeting the IL-6 receptor (e.g., tocilizumab) have been successfully used to treat rheumatoid arthritis, Castleman's disease, and cytokine release syndrome induced by chimeric antigen receptor (CAR)-T cell therapy, and have recently been explored for managing cytokine storms in patients with severe COVID-19[10, 11]. Therefore, a deeper understanding of IL-6 signaling mechanisms is of great significance for developing more specific anti-inflammatory and anti-tumor strategies.
-6%20signal%20pathway%20cascades_.png)
Fig. 2 Known molecules involve in interleukin (IL)-6 signal pathway cascades. Schematic representation of the functional organization of IL-6 receptor and its three downstream transduction. IL-6 cytokine yields its biological effects via two receptors: mgp130 (membrane-bound gp130) and mgp80 (membrane-bound gp80). Each receptor can interact with Janus kinase (JAK) directly. The three pathways all needed JAK and its phosphorylation[13].
04 IL-7: regulation of lymphocyte development and homeostasis
IL-7 is a pleiotropic cytokine essential for both the development and homeostatic maintenance of lymphocytes, and its expression levels can be accurately measured using IL-7 elisa In mice, IL-7 signaling is critical for B lymphopoiesis, promoting the survival, proliferation, and differentiation of progenitor cells; its absence leads to a severe block at the pro-B cell stage[14, 15]. For T cells, IL-7 is indispensable for thymopoiesis, supporting early thymocyte expansion, and continues to play a central role in the periphery by maintaining naive T cell survival and driving lymphopenia-induced proliferation[16]. Mechanistically, IL-7 upregulates anti-apoptotic proteins such as Bcl-2 and Mcl-1 via the JAK/STAT5 pathway, thereby preventing apoptosis[17]. Moreover, in humans, circulating IL-7 levels dynamically respond to T cell depletion: human immunodeficiency virus (HIV)-infected patients and chemotherapy recipients show strong inverse correlations between IL-7 and CD4+ T cell counts, and recovery of CD4 counts is accompanied by a decline in IL-7[18]. Conversely, patients with idiopathic CD4 lymphopenia often lack this inverse relationship, suggesting that impaired IL-7 responsiveness may contribute to their homeostatic defect. These findings collectively position IL-7 as a master regulator of lymphocyte development and homeostasis, with therapeutic implications for immune reconstitution.
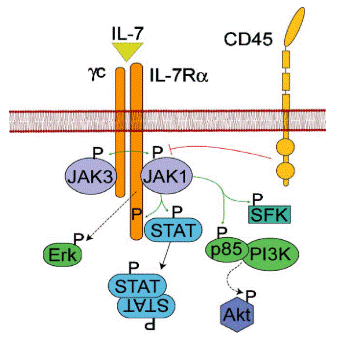
Fig. 3 A schematic view of the IL-7R pathway. IL-7 induces dimerization of the IL-7Rα and γc chains, which subsequently leads to the phosphorylation and activation of JAKs. CD45 can dephosphorylate JAK and may function to limit IL-7R signals. Green arrows represent phosphorylation events. The red line represents dephosphorylation and the broken lines indicate a multi-step pathway leading to activation[14].
05 IL-8: chemotactic function and neutrophil recruitment
IL-8 (also known as CXCL8) is a representative member of the CXC chemokine family, with the primary function of specifically chemoattracting and activating neutrophils, and IL-8 elisa is commonly used to detect its expression in biological samples. Under inflammatory stimulation (e.g., bacteria, viruses, IL-1β, TNF-α), a variety of cells, including epithelial cells, gland duct cells, and macrophages, can synthesize and secrete IL-8. IL-8 binds to CXCR1/CXCR2 receptors on the surface of neutrophils, triggering cytoskeletal rearrangement and directional migration, thereby recruiting neutrophils from the bloodstream to the site of inflammation. Notably, IL-8 not only initiates chemotaxis but also induces migrated neutrophils themselves to produce and release IL-8, forming a positive feedback amplification loop that significantly enhances the local inflammatory response. Taking chronic sinusitis as an example, IL-8 levels in the nasal discharge of patients with this disease can exceed 20 nmol/L, which is approximately ten times the concentration required for maximal chemotactic activity in vitro. Immunohistochemistry and in situ hybridization have shown that nasal gland duct cells, epithelial cells, and neutrophils that have exuded into the sinus cavity all highly express IL-8 and its mRNA[19]. This autocrine and paracrine mechanism drives continuous neutrophil accumulation, leading to the release of proteases and superoxides, impairing the mucociliary function of the mucosa, and resulting in purulent secretion retention and chronic inflammation. Therefore, IL-8 plays a central role in chemotaxis and positive-feedback regulation in neutrophil-dominated inflammation, such as chronic sinusitis and diffuse panbronchiolitis.
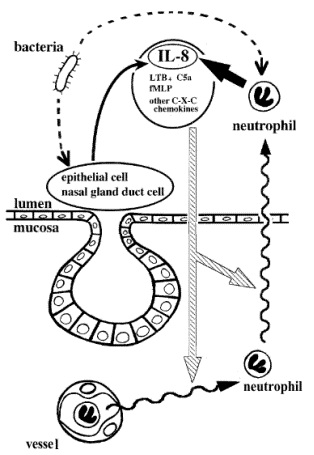
Fig. 4 Hypothesis of neutrophil recruitment in chronic sinusitis. Neutrophil chemotactic substances including IL-8 released from nasal gland duct cells and epithelial cells (thin solidarrow) initiate neutrophil migration (striped arrows) out of vessels to the lumenal side and eventually into sinus effusion (wavy arrows). Emigrated neutrophils produce and secrete IL-8 (thick solid arrow), which further induces neutrophil migration and accumulation in sinus effusion. Bacteria and bacterial components may stimulate neutrophils, epithelial cells, and nasal gland duct cells to secrete IL-8 (dotted arrows). Accumulated neutrophils in sinus effusion probably release proteases and superoxides, which impair mucociliary function of the paranasal epithelium, and this causes retention of sinus effusion. As a result, the whole inflammatory process is prolonged. LTB, Leukotriene B; fMLf, N-formyl-methionyl-leucyl-phenylalanine.
06 Measurement of IL levels using ELISA and AptplexTM assays
The quantitative measurement of interleukin (IL) levels is primarily performed using traditional enzyme-linked immunosorbent assay (ELISA) and emerging multiplex bead array technologies, with multiplex elisa being an efficient approach for simultaneous detection of multiple cytokines. As the "gold standard," ELISA offers high sensitivity, good reproducibility, and extensive validation, but it can only detect one cytokine per assay, requires large sample volumes (e.g., approximately 1,100 μL of serum to measure four cytokines), and has a relatively high cost (about $61.53 per sample)[20]. In contrast, multiplex technologies can simultaneously quantify up to 13 or more cytokines in a single small-volume sample (25-50 μL), at a lower cost (about $14.40 per sample for four cytokines), and with a broader dynamic range[20, 21]. However, multiplex assays in serum/plasma samples may show significant discrepancies compared to ELISA: although most studies report good correlations (often r > 0.85), absolute concentrations often differ, and multiplex assays are more susceptible to matrix effects, antibody cross-reactivity, and sample processing methods (serum vs. plasma)[21, 22]. For example, Wong et al. (2008) found that only IL-4, IL-6, and IL-8 showed significant correlations between serum and plasma, while most ILs had low detection rates in serum (e.g., IL-2, IL-5, and IL-13 had <50% detectability). Therefore, method selection should be based on the study objectives: if high-precision single-analyte validation is needed, ELISA is preferred; if multi-analyte profiling with limited sample volume is required, multiplex assays offer advantages, but key findings should be confirmed by ELISA using identical antibody pairs and consistent sample processing protocols.
07 Crosstalk between ILs and macrophage polarization
IL family members precisely regulate the M1/M2 polarization balance of macrophages through complex signaling networks, thereby playing critical roles in infection, inflammation, tissue repair, and tumor immunity, which is the core of il and macrophage polarization. ILs that induce M1 polarization mainly include IL-2, Interleukin-12 (IL-12), and Interleukin-21 (IL-21), and are typically associated with Th1-type immune responses. For example, IL-2 activates the JAK1/3-STAT5 pathway to promote CD8+ T cell and NK cell activation, while also driving macrophage differentiation toward the M1 phenotype, enhancing antigen presentation and the production of pro-inflammatory cytokines (TNF-α, IL-1β, IL-12, IL-23)[23]. IL-12, mainly produced by activated dendritic cells and M1 macrophages themselves, induces IFN-γ release through STAT4 signaling, forming a positive feedback loop that further consolidates M1 polarization and enhances anti-tumor immunity[24]. IL-21 can reprogram M2 macrophages into the M1 phenotype and exhibits superior anti-tumor activity over IL-2 in tumor models.ILs that promote M2 polarization include IL-4, Interleukin-13 (IL-13), Interleukin-33 (IL-33), and the immunosuppressive Interleukin-10 (IL-10). IL-4 and IL-13 are classic inducers of M2 polarization. They bind to the IL-4Rα receptor, activate the JAK-STAT6 pathway, upregulate M2 markers such as arginase-1 (Arg-1), CD206, FIZZ1, and Ym1, and promote the secretion of anti-inflammatory cytokines IL-10 and transforming growth factor-β (TGF-β), thereby participating in parasite clearance, tissue repair, and fibrosis[24, 25]. Notably, the IL-4/IL-13 axis can also induce KLF4 expression via STAT6, which further cooperates with STAT6 to enhance M2 gene transcription. IL-33 amplifies M2 polarization through the ST2 receptor and plays dual roles in allergic airway inflammation and the tumor microenvironment[23, 25]. IL-10 promotes M2c subtype polarization via STAT3 signaling, inhibits pro-inflammatory cytokine production, and enhances apoptotic cell clearance. Some ILs have dual roles: IL-6 can both synergize with IL-4 to enhance M2 polarization (through STAT3 and BATF) and support M1 responses in acute inflammation. IL-8 (CXCL8), mainly secreted by tumor-associated macrophages (TAMs), promotes M2 polarization and cancer stem cell self-renewal.
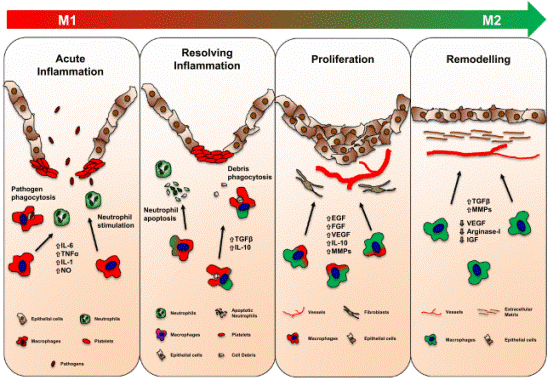
Fig. 5 Role of macrophages in tissue repair. During the different phases of tissue repair, macrophages undergo dynamic changes, switching their phenotype from an M1- to an M2-phenotype.
08 IL-mediated modulation of Treg/Th17 balance
ILs play a critical role in regulating the Treg/Th17 balance, profoundly influencing immune tolerance and inflammatory responses. In patients with obstructive sleep apnoea syndrome (OSAS), levels of the pro-inflammatory cytokines IL-6 and IL-17 are significantly elevated, while the anti-inflammatory cytokine TGF-β1 is decreased, leading to an increased proportion of Th17 cells and a decreased proportion of Treg cells. This disrupts immune tolerance and exacerbates systemic inflammation[26]. Similarly, in inflammatory bowel disease (IBD), a high-calorie diet induces IL-6, which inhibits Treg differentiation and promotes the differentiation of naive CD4⁺ T cells into Th17 cells, whereas IL-10 exerts anti-inflammatory effects through metabolic reprogramming[27]. Moreover, IL-17 has a dual role in the intestinal mucosa: although it can exacerbate inflammation, under certain circumstances (e.g., when derived from γδ T cells) it helps maintain epithelial barrier integrity. In summary, IL-mediated Treg/Th17 imbalance,characterized by the upregulation of IL-6 and IL-17 and the relative deficiency of TGF-β1 and IL-10,is a core mechanism underlying the loss of immune tolerance and the shift in inflammatory cell proportions in inflammatory diseases.
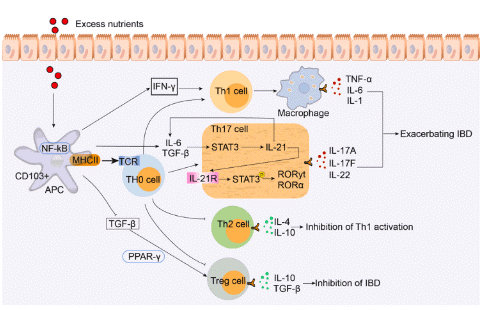
Fig. 6 High-calorie diets maintain an inflammatory environment to promote Th17 cell differentiation and inhibit Treg cell differentiation to worsen the incidence of inflammatory bowel disease. IBD: inflammatory bowel disease.
In summary, IL-1, IL-6, IL-7, and IL-8 play distinct yet interconnected roles in immune regulation. IL-1 and IL-6 serve as central pro-inflammatory mediators: IL-1 activates the inflammasome and NF-κB pathway, while IL-6 utilizes classic and trans-signaling to drive acute-phase responses and chronic inflammation. IL-7 is indispensable for lymphocyte development and peripheral homeostasis, supporting T and B cell survival via the JAK/STAT5 pathway. IL-8 functions as a key chemokine that directs neutrophil chemotaxis and can amplify inflammation through positive feedback loops. Beyond their individual actions, these interleukins critically modulate two major immune axes: macrophage polarization (M1/M2 balance) and the Treg/Th17 equilibrium. Dysregulation of these axes,such as elevated IL-6 and IL-17 alongside reduced TGF-β1 and IL-10,underlies the loss of immune tolerance in autoimmune and chronic inflammatory diseases. Consequently, targeting IL-1 (e.g., anakinra, canakinumab), IL-6 (e.g., tocilizumab), or their downstream pathways has proven therapeutically beneficial. A deeper understanding of the crosstalk among these interleukins will continue to inform the development of precise immunotherapies for inflammation, autoimmunity, and cancer.
Elabscience® Quick Overview of Popular Products:
Table 1. Research Tools for diabetes disease
|
Cat. No. |
Product Name |
|
MIH002N |
EasySortTM Human CD4+ T Cell Isolation Kit |
|
MIH008N |
EasySort™ Human Naïve CD8+ T Cell Isolation Kit |
|
MIM005N |
EasySort™ Mouse NK Cell Isolation Kit |
|
XJH001 |
Human Th1/Th2 Flow Cytometry Staining Kit |
|
XJM004 |
RAW 264.7 Polarized M1 Macrophage Induction and Identification Kit |
|
E-EL-H0088 |
Human IL-1α(Interleukin 1 Alpha) ELISA Kit |
|
CQH015 |
CellaQuant™ Human IL-1β (Interleukin 1 Beta) ELISA Kit |
|
E-EL-H0100 |
Human IL-3(Interleukin 3) ELISA Kit |
|
E-HSEL-M0002 |
High Sensitivity Mouse IL-4 (Interleukin 4) ELISA Kit |
|
CQH012 |
CellaQuant™ Human IL-5 (Interleukin 5) ELISA Kit |
|
E-HSEL-H0003 |
High Sensitivity Human IL-6 (Interleukin 6) ELISA Kit |
|
E-EL-H0648 |
Human IL-7(Interleukin 7) ELISA Kit |
|
CQH004 |
CellaQuant™ Human IL-8 (Interleukin 8) ELISA Kit |
|
E-EL-H0180 |
Human IL-9(Interleukin 9) ELISA Kit |
|
E-HSEL-H0005 |
High Sensitivity Human IL-10 (Interleukin 10) ELISA Kit |
|
CQH019 |
CellaQuant™ Human IL-12 (Interleukin 12) ELISA Kit |
|
E-EL-H0104 |
Human IL-13(Interleukin 13) ELISA Kit |
|
E-CL-H0104 |
Human IL-17 (Interleukin 17) CLIA Kit |
|
E-EL-H1846 |
Human PTGS2/COX-2(Prostaglandin Endoperoxide Synthase 2) ELISA Kit |
|
E-EL-H0109 |
Human TNF-α(Tumor Necrosis Factor Alpha) ELISA Kit |
|
E-EL-H1587 |
Human TGF-β2(Transforming Growth Factor Beta 2) ELISA Kit |
|
MPA019 |
Aptplex™ Human Cytokine 6-Plex Panel |
|
MPD012 |
Aptplex™ Human Cytokine 13-Plex Panel (C) |
References:
[1] Nicod, L.P., Cytokines I. 1993.
[2] Mizel, S.B., The interleukins1. The FASEB Journal, 1989. 3(12): p. 2379-2388.
[3] Dinarello, C.A., Immunological and Inflammatory Functions of the Interleukin-1 Family. Annual Review of Immunology, 2009. 27(1): p. 519-550.
[4] Sims, J.E. and D.E. Smith, The IL-1 family: regulators of immunity. Nature Reviews Immunology, 2010. 10(2): p. 89-102.
[5] Axel Weber, P.W., Michael Kracht, Interleukin-1 (IL-1) Pathway. 2010.
[6] Mantovani, A., et al., Interleukin-1 and Related Cytokines in the Regulation of Inflammation and Immunity. Immunity, 2019. 50(4): p. 778-795.
[7] Dinarello, C.A., Overview of the interleukin-1 family of ligands and receptors. Seminars in Immunology, 2013. 25(6): p. 389-393.
[8] Su, H., C.-T. Lei, and C. Zhang, Interleukin-6 Signaling Pathway and Its Role in Kidney Disease: An Update. Frontiers in Immunology, 2017. 8.
[9] Rose-John, S., IL-6 Trans-Signaling via the Soluble IL-6 Receptor: Importance for the Pro-Inflammatory Activities of IL-6. International Journal of Biological Sciences, 2012. 8(9): p. 1237-1247.
[10] Rose-John, S., Interleukin-6 signalling in health and disease. F1000Research, 2020. 9.
[11] Jones, S.A. and B.J. Jenkins, Recent insights into targeting the IL-6 cytokine family in inflammatory diseases and cancer. Nature Reviews Immunology, 2018. 18(12): p. 773-789.
[12] Hirano, K.I.T., IL-6 in autoimmune disease and chronic inflammatory proliferative disease. 2002.
[13] Luo, Y. and S.G. Zheng, Hall of Fame among Pro-inflammatory Cytokines: Interleukin-6 Gene and Its Transcriptional Regulation Mechanisms. Frontiers in Immunology, 2016. 7.
[14] Milne, C.D. and C.J. Paige, IL-7: A key regulator of B lymphopoiesis. Seminars in Immunology, 2006. 18(1): p. 20-30.
[15] Ceredig, R. and A.G. Rolink, The key role of IL-7 in lymphopoiesis. Seminars in Immunology, 2012. 24(3): p. 159-164.
[16] Carrette, F. and C.D. Surh, IL-7 signaling and CD127 receptor regulation in the control of T cell homeostasis. Seminars in Immunology, 2012. 24(3): p. 209-217.
[17] Sun-Kyeong Lee, C.D.S., Role of interleukin-7 in bone and T-cell homeostasis. 2005.
[18] Terry J. Fry, E.C., Judith Falloon, Michael M. Lederman, David J. Liewehr, John Spritzler, Seth M. Steinberg, Lauren V. Wood, Robert Yarchoan, Judy Zuckerman, Alan Landay, and Crystal L. Mackall, Apotential role for interleukin-7 in T-cell homeostasis. 2001.
[19] Hideaki Suzuki, M., Yuichi Takahashi, MD, Hideya Wataya, MD, Katsuhisa Ikeda, MD, Seiichiro Nakabayashi, MD, Akira Shimomura, MD, and Tomonori Takasaka, MD, Mechanism of neutrophil recruitment induced by IL-8 in chronic sinusitis. 1996.
[20] Sean X. Leng, J.E.M., Jeremy D. Walston, Dongxu Xie, Neal S. Fedarko, and George A. Kuchel, ELISA and Multiplex Technologies for Cytokine Measurement in Inflammation and Aging Research. 2008.
[21] Wong, H.-L., et al., Reproducibility and Correlations of Multiplex Cytokine Levels in Asymptomatic Persons. Cancer Epidemiology, Biomarkers & Prevention, 2008. 17(12): p. 3450-3456.
[22] Mohamed F. Elshal, P.D.a.J.P.M.J., PhD, Multiplex Bead Array Assays: Performance Evaluation and Comparison of Sensitivity to ELISA. 2006.
[23] Mortezaee, K. and J. Majidpoor, Roles for macrophage-polarizing interleukins in cancer immunity and immunotherapy. Cellular Oncology, 2022. 45(3): p. 333-353.
[24] Tugal, D., X. Liao, and M.K. Jain, Transcriptional Control of Macrophage Polarization. Arteriosclerosis, Thrombosis, and Vascular Biology, 2013. 33(6): p. 1135-1144.
[25] Sica, A., et al., Macrophage polarization in pathology. Cellular and Molecular Life Sciences, 2015. 72(21): p. 4111-4126.
[26] Ye, J., et al., The Treg/Th17 Imbalance in Patients with Obstructive Sleep Apnoea Syndrome. Mediators of Inflammation, 2012. 2012: p. 1-11.
[27] Zhang, S., et al., Metabolic regulation of the Th17/Treg balance in inflammatory bowel disease. Pharmacological Research, 2024. 203.



