Chimeric Antigen Receptor T-cell Immunotherapy (CAR-T therapy) has exhibited remarkable efficacy in the treatment of hematological malignancies, including acute leukemia and non-Hodgkin lymphoma. Thus it is recognized as one of the most promising therapeutic modalities for cancer. However, its application in solid tumors remains challenging, primarily due to multiple obstacles such as antigen heterogeneity, the immunosuppressive tumor microenvironment, and T cell exhaustion.
Compared with bulk T cells (i.e., total T cells), naive T cells are regarded as the "gold-standard" starting population in CAR-T research. This is mainly attributed to the fact that naive T cells represent a relatively undifferentiated yet highly potent T-cell subset, which possesses the potential to overcome the current limitations of CAR-T therapy. In this article, we detail the unique characteristics and advantages of naive T cells, explain the rationale for their preference over bulk T cells in CAR-T manufacturing, and elaborate on the methods and underlying principles for their isolation.
Table of Contents
1. What are naïve T cells?
2. Why CAR-T research prefer naïve T cells rather than total T cells?
3. Introduction to naïve T Cell Isolation
01 What are naïve T cells?
Naïve T cells (Tn) represent a subset of T cells that have not yet been exposed to specific antigens, and they are key components of the immune system in combating new pathogens[1]. In the research of CAR-T cell therapy, the selection of appropriate T cell starting materials is crucial for the efficacy, persistence, and safety of CAR-T products[2]. Different from the cell populations that have been exposed to antigens and differentiated into memory T cells or effector T cells, naïve T cells possess unique biological characteristics, which make them one of the preferred cell sources for CAR-T cell therapy[3].
The main characteristics of naïve T cells (Tn) include the following:
High proliferative potential: Upon stimulation by antigens, naïve T cells exhibit robust proliferative capacity, enabling rapid expansion into a large number of effector T cells and memory T cells[3]. This high proliferative potential is crucial for both the in vitro expansion of CAR-T cells and the in vivo persistence of CAR-T cells[3].
Long lifespan and low exhaustion propensity: Naïve T cells typically have a long lifespan and stronger resistance to immune exhaustion[3]. In CAR-T cell therapy, exhaustion is a major barrier that can lead to functional inactivation of CAR-T cells and treatment failure. Using naïve T cells as the starting material helps generate CAR-T cells with more durable functions and stronger anti-tumor activity[2].
Differentiation pluripotency: Naïve T cells possess the potential to differentiate into multiple T cell subsets, including central memory T cells (TCM) and effector memory T cells (TEM)[1]. Particularly, central memory T cells are considered to play a key role in maintaining long-term immune protection due to their "stem cell-like" properties, which is essential for the long-term survival of CAR-T cells and their sustained anti-tumor effects[3].
02 Why CAR-T research prefer naïve T cells rather than total T cells?
In CAR-T cell research, the preferential selection of naïve T cells (Tn) as starting material, as opposed to total T cells, is primarily aimed at optimizing the therapeutic efficacy, persistence, and safety of the resulting CAR-T product[4,5,6]. This preference is rooted in the distinct biological properties of naïve T cells, which are believed to significantly enhance the success of CAR-T cell therapies.
First, naïve T cells exhibit superior proliferative capacity and differentiation potential. Being antigen-inexperienced, they can undergo extensive expansion following in vitro activation and genetic modification, yielding a larger number of CAR-T cells[4]. This robust proliferative potential is critical for generating sufficient cell numbers for therapy and supports the long-term persistence and sustained anti-tumor activity of CAR-T cells in vivo. Furthermore, naïve T cells possess the ability to differentiate into various T-cell subsets, including central memory T cells (Tcm) and effector memory T cells (Tem)[4]. Tcm, in particular, are known for their stem cell-like properties, enabling long-term survival and maintenance of immunological memory, which is crucial for durable CAR-T responses. Studies have shown that CAR-T products enriched with a Tcm-like phenotype typically exhibit improved anti-tumor efficacy[4,6]. In contrast, the total T-cell population comprises a heterogeneous mix of cells at various differentiation stages, including a significant proportion of terminally differentiated or exhausted T cells[7]. These subsets show limited expansion potential and reduced persistence, which can compromise the overall potency of the CAR-T product[8].
Second, naïve T cells demonstrate enhanced resistance to exhaustion and greater in vivo persistence[4]. During tumor eradication, CAR-T cells face an immunosuppressive tumor microenvironment and persistent antigen exposure, leading to T cell exhaustion, which is characterized by loss of effector function, reduced proliferation, and upregulation of inhibitory receptors such as PD-1[7,8]. Naïve T cells, and the Tcm derived from them, are inherently more resilient to such exhaustion[4]. For instance, strategies to downregulate PD-1 expression in CAR-T cells have been shown to enhance anti-tumor efficiency, correlating with the preservation of a memory-like phenotype and reduced exhaustion[7].
Additionally, from the perspective of T-cell quality, many patients eligible for CAR-T therapy have undergone multiple prior lines of chemotherapy or other preconditioning regimens. These treatments can compromise the endogenous T-cell pool, resulting in reduced T-cell numbers, functional impairment, or an exhausted state[4]. In such cases, using therapy naïve T cells, even if not strictly a pure naïve population, as the starting material can effectively enhance the potency of the resulting CAR-T product. This approach has demonstrated promising outcomes in patients with B-cell non-Hodgkin lymphoma (B-NHL), underscoring the importance of the "fitness" of the starting T-cell population for CAR-T efficacy[4].
Although total T cells are readily accessible and provide a broad spectrum of T cell subsets for CAR-T manufacturing, their heterogeneity risks incorporating functionally inferior or over-differentiated cells into the final product, potentially impairing expansion, persistence, and anti-tumor activity[8]. Therefore, the precise selection and enrichment of naïve T cells enable researchers and clinicians to develop more potent and durable CAR-T cell products, thereby significantly improving treatment outcomes for cancer patients[4].
03 Introduction to naïve T Cell Isolation
Operationally, T cell selection is typically achieved using antibody-based techniques such as magnetic-activated cell sorting or fluorescence-activated cell sorting (FACS)[3]. FACS can pose challenges related to high cost, time consumption, and potential cell damage. In contrast, label-based magnetic negative selection enables the efficient isolation of highly pure naïve T cells, providing a superior starting population for CAR-T therapy[3]. This precise selection of T cell subsets contributes to enhanced potency and persistence, thereby improving the predictability of therapeutic outcomes[3].
The Elabscience® Human Naïve Pan T Cell Isolation Kit utilizes an indirect magnetic negative isolation strategy for the purification of naïve T cells from PBMCs. By targeting unwanted cells for depletion with biotinylated antibodies and streptavidin beads, the desired naïve T cells remain untouched and unlabeled. This process prevents antibody-induced activation or epitope masking, yielding pristine cells in their native state. Such high-quality starting material ensures more reliable and reproducible results in downstream CAR-T applications, from in vitro differentiation to in vivo adoptive immunotherapy.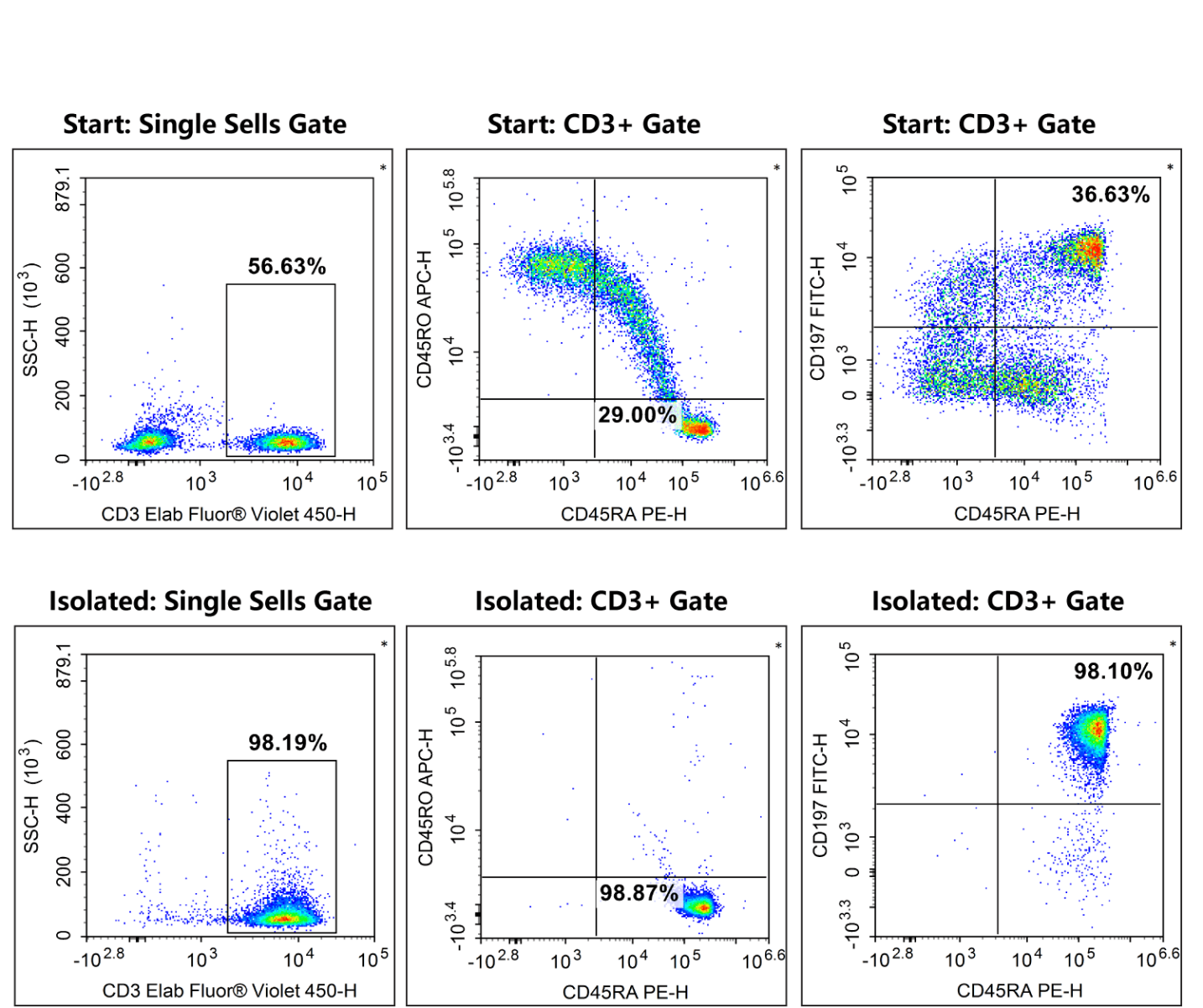
Fig. 1 The purities of human naïve pan T cells (CD3+CD45RA+CD45RO-CD197+) in normal human peripheral blood mononuclear cell samples were 6.0% before sorting and 95.2% after sorting.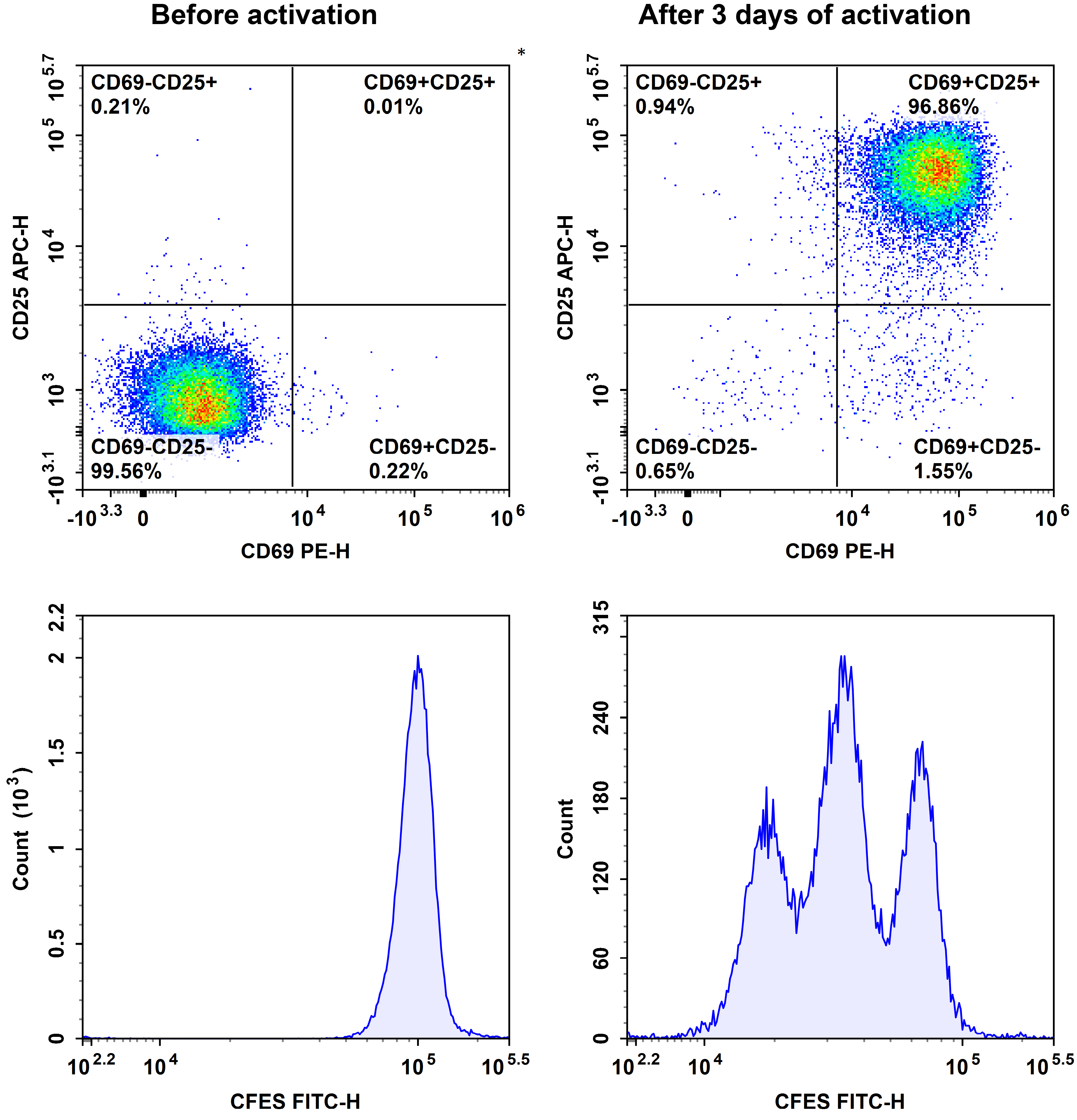
Fig. 2 The purified human naïve pan T cells were stained with CFSE, then activated with Human CD3/CD28 T Cell Activation Beads (MIH001A) for 3 days. The proliferation of T cells before and after activation was detected by CD69, CD25 and CFSE.
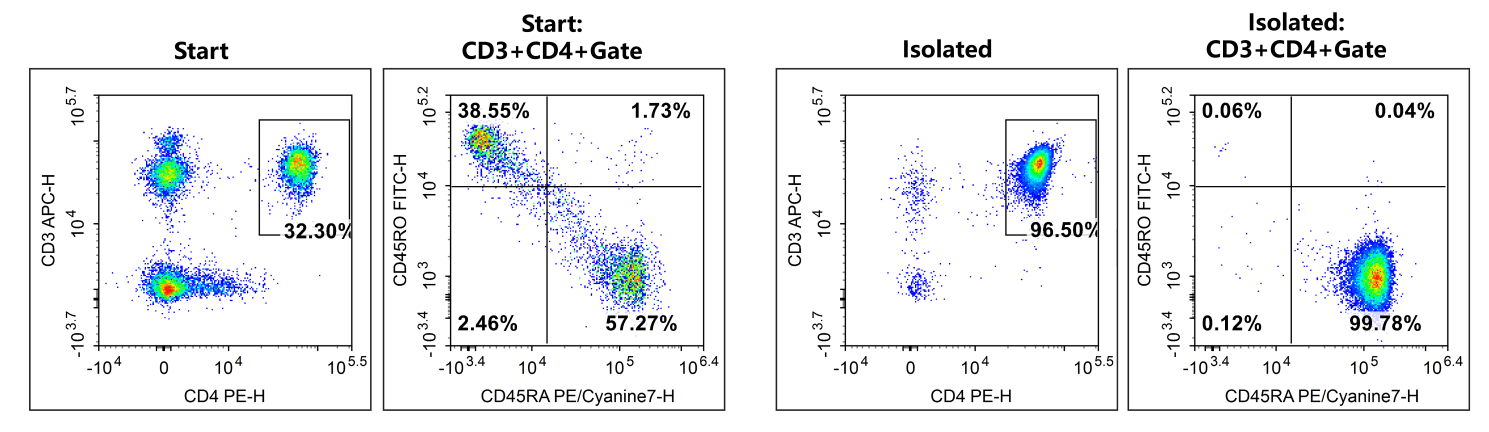
Fig. 3 The purities of human naïve CD4+ T cells (CD3+CD4+CD45RA+CD45RO-) in normal human peripheral blood mononuclear cell samples were 18.49% before sorting and 96.28% after sorting.
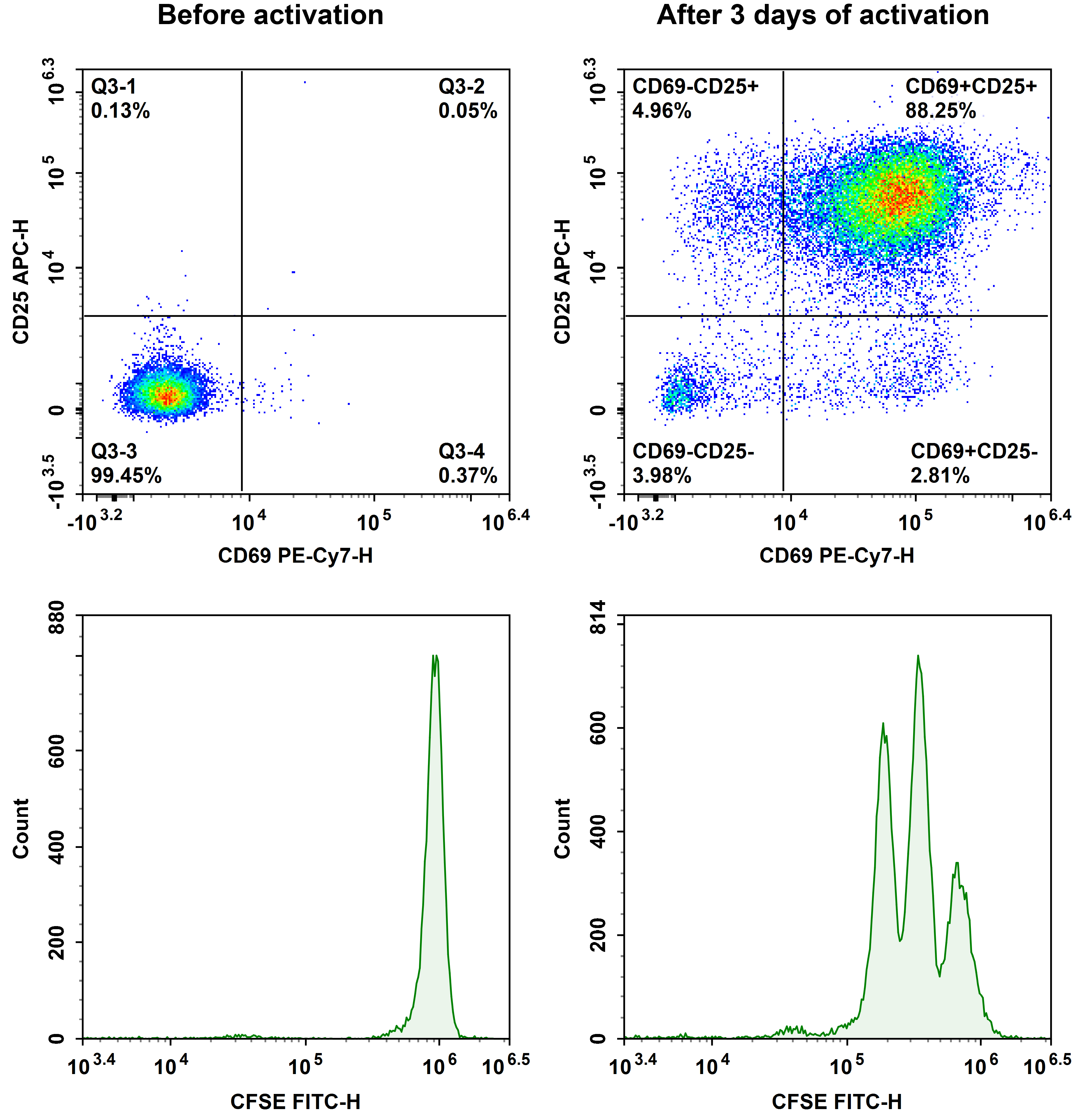
Fig. 4 The purified human naïve CD4+ T cells were stained with CFSE, then activated with Human CD3/CD28 T Cell Activation Beads (MIH001A) for 3days. The proliferation of T cells before and after activation was detected by CD69, CD25 and CFSE.
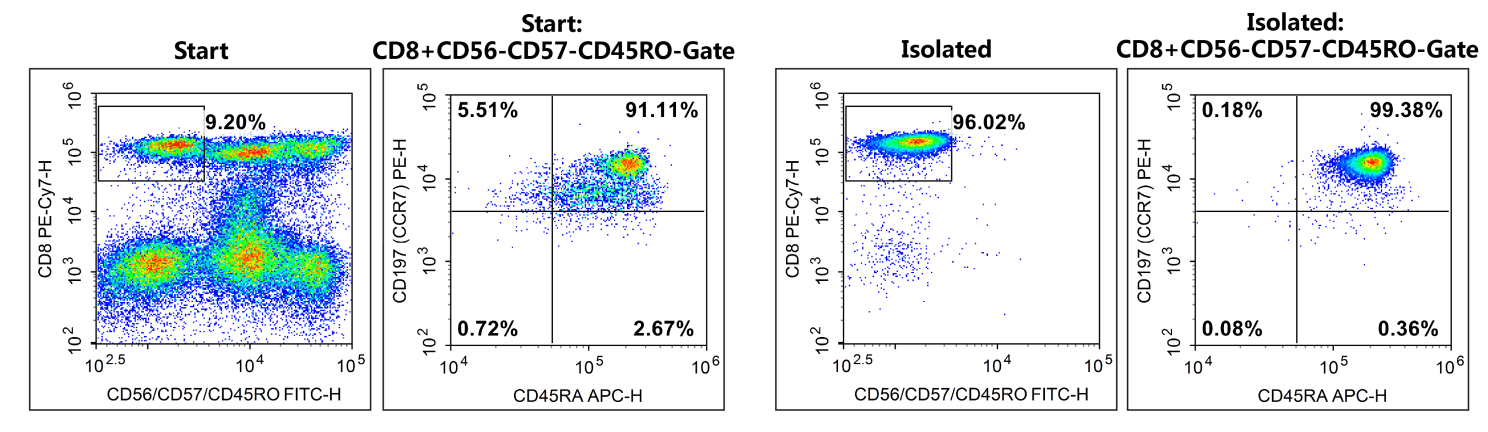
Fig. 5 The purities of Naive CD8+T cells (CD3+CD56-CD5-CD45RO-CD8+CD45RA+CD196+) before and after sorting were analyzed by flow cytometry. The proportions of Naive CD8+T cells before and after sorting were 8.4% and 95.4%, respectively.
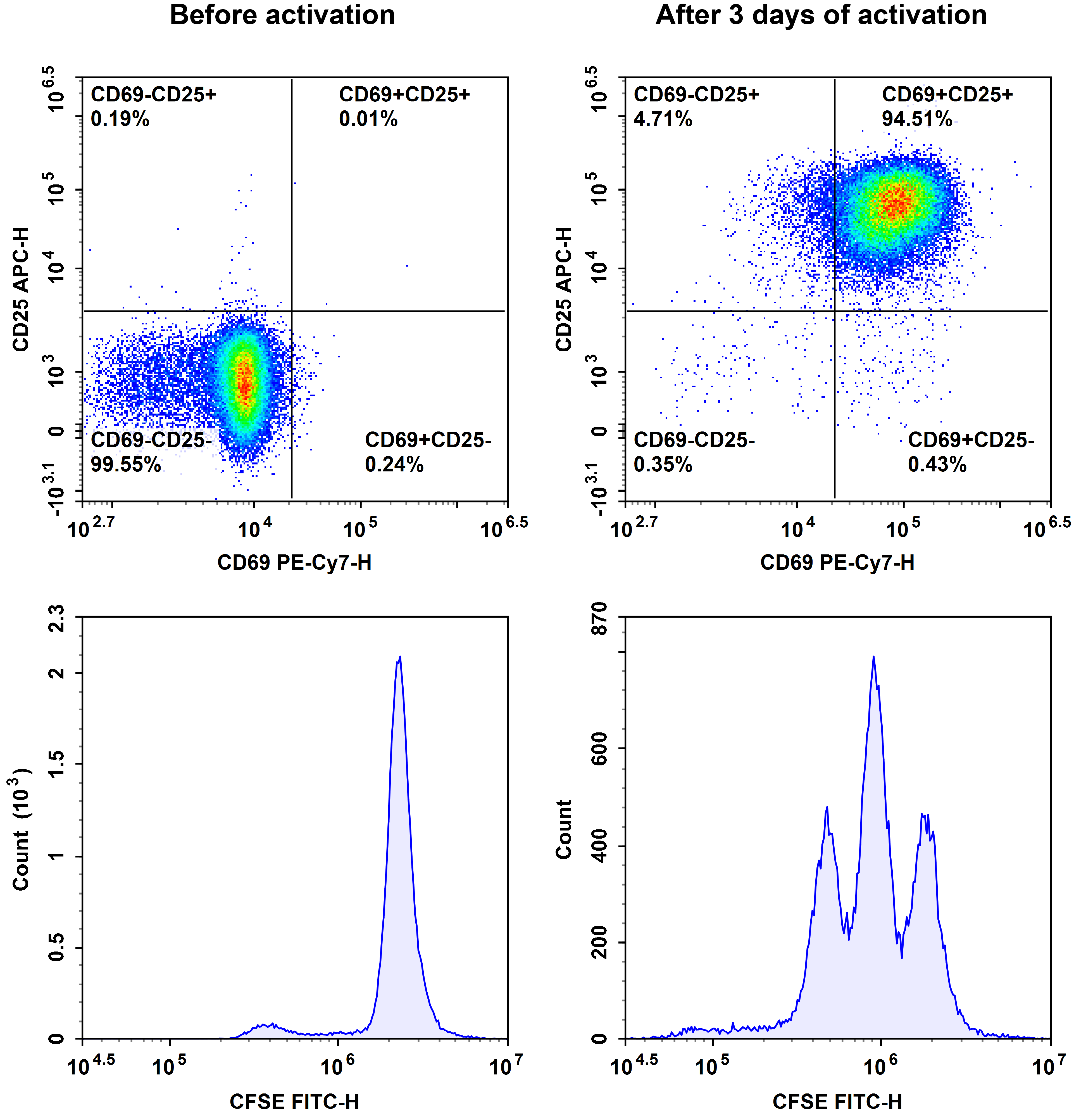
Fig. 6 The purified human naïve CD8+ T cells were stained with CFSE, then activated with Human CD3/CD28 T Cell Activation Beads (MIH001A) for 3days. The proliferation of T cells before and after activation was detected by CD69, CD25 and CFSE.
Core Advantages of Elabscience® EasySort™ Cell Isolation Products:
◆ No need for red blood cell lysis treatment, ensur higher cell viability.
◆ Without stimulation, the target sorted cells are free of antibody and magnetic bead labels, so their cellular state remains unaffected.
◆ High purity: the purity of sorted cells exceeds 95%.
◆ No separation column is needed for the isolation process.
◆ Short experimental time: negative cell sorting can be completed in as fast as 15 minutes.
◆ Applicable for sorting fresh human peripheral blood mononuclear cell (PBMC) samples or cryopreserved PBMC samples.
Elabscience® Quick Overview of Popular Products:
Table 1. Multicolor Panel for Flow Cytometric Analysis of Human T cells
|
Marker |
Clone |
Fluorochrome |
Cat. No. |
Applications |
|
CD3 |
UCHT1 |
PerCP/Cyanine5.5 |
E-AB-F1230J |
Detection of Human T Cell Activation Status |
|
CD69 |
FN50 |
APC |
E-AB-F1138E |
|
|
CD25 |
CHI621 |
PE |
AN00360D |
|
|
HLA-DR |
L243 |
FITC |
E-AB-F1111C |
|
|
CD8 |
OKT-8 |
PE/Cyanine7 |
E-AB-F1110H |
Detection of Human Naive CD8 T Cells |
|
CD45RA |
HI100 |
APC |
E-AB-F1052E |
|
|
CD45RO |
UCHL1 |
FITC |
E-AB-F1139C |
|
|
CD56 |
MY31 |
FITC |
E-AB-F1270C |
|
|
CD57 |
HNK-1 |
FITC |
E-AB-F1067C |
|
|
CD197/CCR7 |
G043H7 |
PE |
E-AB-F1159D |
|
|
DAPI |
/ |
Pacific Blue |
E-CK-A163 |
|
|
CD3 |
OKT-3 |
APC |
E-AB-F1001E |
Detection of Human Naive CD4 T Cells |
|
CD4 |
SK3 |
PE |
E-AB-F1352D |
|
|
CD45RA |
HI100 |
PerCP |
E-AB-F1052F |
|
|
CD45RO |
UCHL1 |
FITC |
E-AB-F1139C |
|
|
CD3 |
OKT-3 |
Elab Fluor®700 |
E-AB-F1001M1 |
Detection of Human Naive/Memory T cells |
|
CD4 |
SK3 |
PerCP/Cyanine5.5 |
E-AB-F1109J |
|
|
CD45RA |
HI100 |
FITC |
E-AB-F1052C |
|
|
CD197/CCR7 |
G043H7 |
PE |
E-AB-F1159D |
Table 2. Reagents for Human T cells Tregs Research
|
Product Name |
Cat. No. |
|
Human PBMC Separation Solution(P 1.077) |
E-CK-A103 |
|
EasySortTM Human Naïve Pan T Cell Isolation Kit |
MIH006N |
|
EasySortTM Human Naïve CD4+ T Cell Isolation Kit |
MIH007N |
|
EasySortTM Human Naïve CD8+ T Cell Isolation Kit |
MIH008N |
|
Human Th1/Th2 Flow Cytometry Staining Kit |
XJH001 |
|
Human Th17 Flow Cytometry Staining Kit |
XJH002 |
|
Cell Stimulation and Protein Transport Inhibitor Kit |
E-CK-A091 |
|
Human CD3/CD28 T Cell Activation Beads |
MIH001A |
|
CFSE Cell Division Tracker Kit |
E-CK-A345 |
|
EasySort™ Human CD3+T Cell Isolation Kit |
MIH001N |
|
Foxp3/Transcription Factor Staining Kit |
E-CK-A108 |
|
Intracellular Fixation/Permeabilization Buffer Kit |
E-CK-A109 |
|
EasySort™-5 Magnet |
EC001 |
|
10×ACK Lysis Buffer |
E-CK-A105 |
For more products and related reagents, click here to view our complete T Cell Sorting product list.
References:
[1] Galletti, G., De Simone, G., Mazza, E. M. C., Puccio, S., Mezzanotte, C., Bi, T. M., Davydov, A. N., Metsger, M., Scamardella, E., Alvisi, G., De Paoli, F., Zanon, V., Scarpa, A., Camisa, B., Colombo, F. S., Anselmo, A., Peano, C., Polletti, S., Mavilio, D., … Lugli, E. (2020). Two subsets of stem-like CD8+ memory T cell progenitors with distinct fate commitments in humans. Nature Immunology, 21(12), 1552–1562. https://doi.org/10.1038/s41590-020-0791-5.
[2] Junkuhn, C., Schiele, P., Busch, D., Polansky-Biskup, J., Künkele-Langer, A., Obermayer, B., Bullinger, L., Frentsch, M., & Na, I.-K. (2024). Utilization of Therapy-Naïve T Cells to Enhance the Efficacy of CAR-T Cells in the Treatment of B-NHL Patients. Blood, 144(Supplement 1), 2027–2027. https://doi.org/10.1182/blood-2024-199124.
[3] Wu, A. Y., Cheng, E. L., Kacherovsky, N., Marking, A., Lin‐Goldstein, A., Heinze, C. M., Salipante, S. J., Jensen, M. C., & Pun, S. H. (2025). Efficient and Traceless Aptamer‐Based Serial Selection of Naïve and Early Memory CD8 T Cells for CAR T Cell Therapy. Advanced Healthcare Materials. https://doi.org/10.1002/adhm.202502930.
[4] Junkuhn, C., Schiele, P., Busch, D., Polansky-Biskup, J., Künkele-Langer, A., Obermayer, B., Bullinger, L., Frentsch, M., & Na, I.-K. (2024). Utilization of Therapy-Naïve T Cells to Enhance the Efficacy of CAR-T Cells in the Treatment of B-NHL Patients. Blood, 144(Supplement 1), 2027–2027. https://doi.org/10.1182/blood-2024-199124.
[5] Wu, A. Y., Cheng, E. L., Kacherovsky, N., Marking, A., Lin‐Goldstein, A., Heinze, C. M., Salipante, S. J., Jensen, M. C., & Pun, S. H. (2025). Efficient and Traceless Aptamer‐Based Serial Selection of Naïve and Early Memory CD8 T Cells for CAR T Cell Therapy. Advanced Healthcare Materials. https://doi.org/10.1002/adhm.202502930.
[6] Galletti, G., De Simone, G., Mazza, E. M. C., Puccio, S., Mezzanotte, C., Bi, T. M., Davydov, A. N., Metsger, M., Scamardella, E., Alvisi, G., De Paoli, F., Zanon, V., Scarpa, A., Camisa, B., Colombo, F. S., Anselmo, A., Peano, C., Polletti, S., Mavilio, D., … Lugli, E. (2020). Two subsets of stem-like CD8+ memory T cell progenitors with distinct fate commitments in humans. Nature Immunology, 21(12), 1552–1562. https://doi.org/10.1038/s41590-020-0791-5.
[7] Ouyang, W., Jin, S.-W., Xu, N., Liu, W.-Y., Zhao, H., Zhang, L., Kang, L., Tao, Y., Liu, Y., Wang, Y., Wang, J., Liu, F., Yu, L., Liu, Z., & Mi, J.-Q. (2024). PD-1 downregulation enhances CAR-T cell antitumor efficiency by preserving a cell memory phenotype and reducing exhaustion. Journal for ImmunoTherapy of Cancer, 12(4), e008429. https://doi.org/10.1136/jitc-2023-008429.
[8] Bulliard, Y., Andersson, B. S., Baysal, M. A., Damiano, J., & Tsimberidou, A. M. (2023). Reprogramming T cell differentiation and exhaustion in CAR-T cell therapy. Journal of Hematology & Oncology, 16(1). https://doi.org/10.1186/s13045-023-01504-7.
[9] Wu, A. Y., Cheng, E. L., Kacherovsky, N., Marking, A., Lin‐Goldstein, A., Heinze, C. M., Salipante, S. J., Jensen, M. C., & Pun, S. H. (2025). Efficient and Traceless Aptamer‐Based Serial Selection of Naïve and Early Memory CD8 T Cells for CAR T Cell Therapy. Advanced Healthcare Materials. https://doi.org/10.1002/adhm.202502930.



