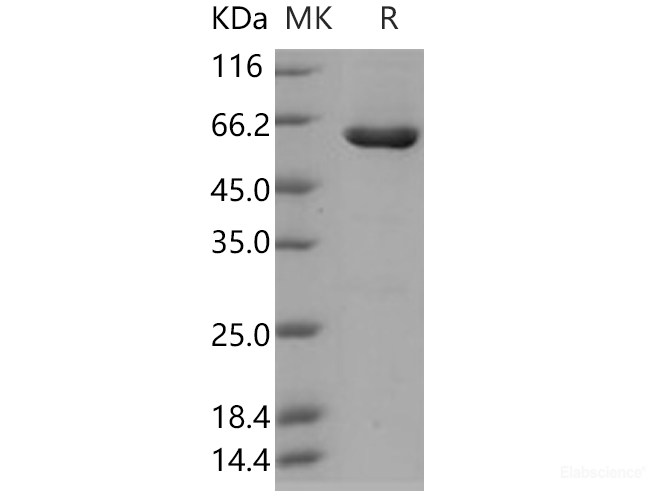
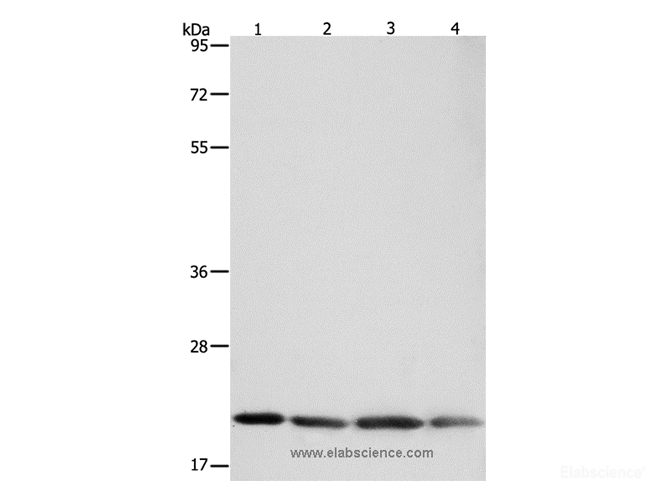
Background
Casein kinase II subunit alpha', also known as CSNK2A2 and CK2A2, is a member of the protein kinase superfamily, Ser/Thr protein kinase family and CK2 subfamily. Casein kinases are operationally defined by their preferential utilization of acidic proteins such as caseins as substrates. The alpha and alpha' chains contain the catalytic site. CSNK2A2 is a tetramer composed of an alpha chain, an alpha' and two beta chains. It is also component of a CK2-SPT16-SSRP1 complex composed of SSRP1, SUPT16H, CSNK2A1, CSNK2A2 and CSNK2B, the complex associating following UV irradiation. Protein kinase casein kinase II (Ck2) is a cyclic-AMP and calcium-independent serine-threonine kinase that is composed of two catalytic subunits (alpha and alpha') and two regulatory beta-subunits. Ck2 is not a casein kinase in vivo, but over 100 substrates are known. The highly conserved amino acid sequences of its subunits and their broad expression suggest that Ck2 may have a fundamental role in cell function. Ck2 has been implicated in DNA replication, regulation of basal and inducible transcription, translation and control of metabolism. The Ck2alpha and Ck2alpha' isoforms (products of the genes Csnk2a1 and Csnk2a2, respectively) are highly homologous, the reason for their redundancy and evolutionary conservation is unknown. CSNK2A2 may be a candidate gene for these inherited syndromes.